issue contents
September 2021 issue
Cover illustration: The OXA-48-like carbapenemase OXA-436. Lund et al. [(2021), Acta Cryst. F77, 312–318] explore the reasons why the antibiotic-resistance enzyme OXA-436 has a higher enzymatic activity at physiological temperatures than its its more widespread homolog OXA-48.
research communications
Several crystal structures are analyzed of proteins that form the shells of bacterial microcompartment organelles. These new structures are from microcompartments where the metabolic substrate choline diffuses across the shell-protein pore.
PDB references: CmcA, 7mgp; CmcB, 7mpw; CmcB, E35G mutant, 7mn4; CmcC, 7mpv; CmcC, E35G mutant, 7mpx; CutN, 7mmx
Open  access
access
 access
accessSite 2 of the Yersinia pestis substrate-binding protein YfeA is a dynamic surface metal-binding site
The Yersinia pestis substrate-binding protein YfeA contains two polyspecific metal-binding sites, and site 2 is capable of collaborating with other protein molecules for inter-protein metal coordination. The inter-protein metal coordination can occur by multiple coordination geometries and may be flexible enough to enable metal transfer to multiple metal-binding proteins.
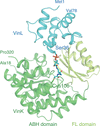
Protein–protein interaction between the acyltransferase VinK and the carrier protein VinL is critical for substrate transfer in vicenistatin biosynthesis. The crystal structure of the VinK–VinL covalent complex formed with a pantetheine cross-linking probe has been determined.
Crystal structures of glycogen phosphorylase in the free T-state and R-state and in the R-state in complex with the allosteric activators IMP and AMP are reported at improved resolution.
Open  access
access
 access
accessPossible mechanisms are demonstrated to explain why the antibiotic-resistance enzyme OXA-436 has higher catalytic rates towards last-resort antibiotics under physiological conditions compared with its more widespread homolog OXA-48.
PDB reference: OXA-48-like β-lactamase OXA-436, 7oda
methods communications
Download citation


Download citation


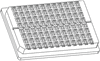
A novel device has been designed that could be used as both an in situ macroplate and in situ microplate for high-throughput crystal screening and in situ data collection.
PDB reference: lysozyme, 7f26


 journal menu
journal menu
















![[publBio]](/logos/publbio.gif)





