issue contents
Halogen, chalcogen, pnictogen and tetrel bonds: structural chemistry and beyond
Edited by Lee Brammer, Anssi Peuronen and Thomas M. Roseveare
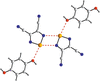
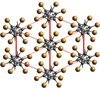
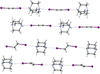
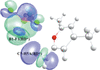
This virtual special issue of Acta Crystallographica Section C: Structural Chemistry brings together 12 articles that cover a variety of aspects associated with halogen, chalcogen, pnictogen and tetrel bonds. There are articles that focus on structure, bonding and bond strength, articles that investigate co-operation or competition between different classes of interactions, articles that explore the relationship between these interactions and chemical or physical properties of the compounds and materials involved, and, although all articles involve characterization by single-crystal X-ray diffraction, a number have a prominent focus on other experimental techniques or are combined with interpretation from theoretical calculations. Halogen bonding and chalcogen bonding dominate these articles, consistent with the wider literature.

Cover illustration: A snapshot of current progress in the field of halogen bonds, chalcogen bonds, pnictogen bonds, tetrel bonds and other σ-hole interactions. Courtesy of Brammer et al. [Acta Cryst. (2023), 79, 204–216].
 access
access



 access
access





 access
access



 access
access



 access
access

 access
access
 journal menu
journal menu