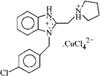
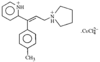
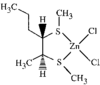
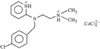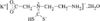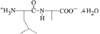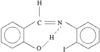issue contents
June 1997 issue
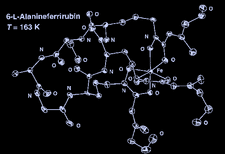
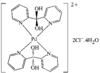
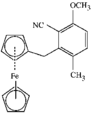
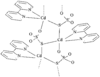
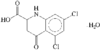
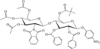
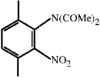
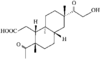
Cover illustration: 6-L-Alanineferrirubin, a Ferrichrome-type Siderophore from the Fungus Aspergillus ochraceous, see Hossain, Jalal and van der Helm, pages 716-718. Displacement ellipsoids are shown at the 50% probability level and H atoms and water molecules have been omitted for clarity.
cif-access (metal-organic compounds)
Download citation


Download citation


cif-access (organic compounds)
Download citation


Download citation


Download citation


Download citation


inorganic compounds
Download citation


Download citation


Rietveld refinement of X-ray powder data shows that these Jahn–Teller spinels contain flattened CuO4 tetrahedra and regular CrO6 or RhO6 octahedra.
Download citation


Download citation


The structure of [CoCl(NH3)5][Ru(NO2)4(OH)(NO)] consists of octahedral [CoCl(NH3)5]2+ and [Ru(NO2)4(OH)(NO)]2− ions held together via a three-dimensional network of inter- molecular O—H⋯O, N-H⋯O and N-H⋯Cl hydrogen bonds.
Download citation


Download citation


The structure of [Co(NH3)6](S2O3)Cl.H2O consists of discrete ions and water molecules of hydration linked by weak hydrogen bonds and interionic contacts.
Download citation


Download citation


Cs2Au2I6 has a distorted perovskite structure, isomorphous with the Wells salt Cs2Au2Cl6. The interatomic distances in Cs2Au2I6 show that the mixed-valence state of the iodide is not the same as that of the chloride.
Download citation


Download citation


Mg(HSeO3)2 contains MgO6 octahedra and strongly hydrogen-bonded dimers of trigonal pyramidal SeO2OH− ions in an NaCl-like arrangement.
Download citation


Download citation


SmMo8O14 is isostructural with NdMo8O14. The structure contains cis-edge-sharing bi-face-capped octahedral Mo8 clusters arranged in layers. The O-atom framework is derived from the close-packing layer sequence ABAC. The Sm3+ ions are surrounded by 12 O atoms forming a distorted cuboctahedron.
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


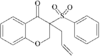
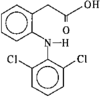
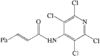
organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


addenda and errata
Free 

Erratum to Acta Cryst. (1996), C52, 3207–3210.


 journal menu
journal menu