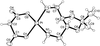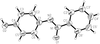issue contents
June 2000 issue
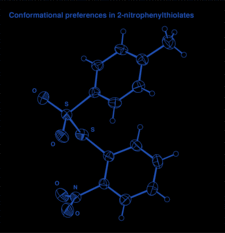
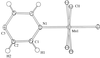
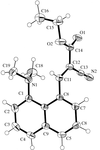
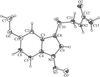
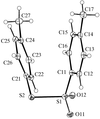
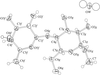
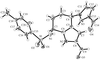
Cover illustration: Conformational preferences in 2-nitrophenylthiolates: S-(2-nitrophenyl) 4-toluenethiosulfonate. Ellipsoids are plotted at the 30% probability level and H atoms are shown as small spheres of arbitrary size. See Low, Glidewell & Wardell [Acta Cryst. C56, 689-691].
inorganic compounds
Download citation


Download citation


Gd2OSe2 is the first rare-earth monooxygen diselenide to be characterized structurally. It is isostructural with the low-pressure form of Dy2OS2. Its structure comprises the close packing of Gd-centered bicapped trigonal prisms and Gd-centered distorted octahedra.
Download citation


Download citation


CsTiP2O7 crystallizes in the KAlP2O7 structure type. It has a three-dimensional tunnel structure built by the corner-sharing of distorted TiO6 octahedra and P2O7 pyrophosphate groups. The Cs+ cations are in the tunnels.
Download citation


Download citation


The new structure of La2(SeO3)3 consists of LaO10 polyhedra and SeO3 pyramids sharing faces, edges and vertices. The three-dimensional structure contains pseudo-channels associated with the SeIV lone pairs.
Download citation


Download citation


The structure of monoclinic dipotassium dichromate consists of K+ cations and Cr2O72− dimers.
Download citation


Download citation


The structure of RbLi2Ga2(BO3)3 is an ordered derivative of KZn(BO3)3. It exhibits a three-dimensional framework of BO3 triangles, and LiO4 and GaO4 tetrahedra that isolates the Rb atoms in channels extending along the b axis.
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


electronic papers (inorganic compounds)
Download citation


Download citation


Precise X-ray structure determinations of K3Na[FeCl6] have been performed at 293, 84 and 9.5 K.
Download citation


Download citation


The title structure was redetermined at 120 K, confirming the hydrogen-bond pattern found at room temperature, but showing the librational corrections of bond lengths to be unrealistic. The octahedral coordination of Mg atoms is slightly distorted due to hydrogen bonding.
Download citation


Download citation


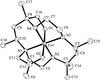
Crystals of KBa4Bi3O have been obtained from melting of the elements. The structure contains Bi24− dumbbells [Bi—Bi = 3.113 (3) Å] and isolated Bi3− anions coordinated by K and Ba atoms. O atoms lie at the centres of Ba tetrahedra.
electronic papers (metal-organic compounds)
Download citation


Download citation


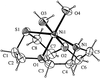
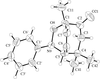
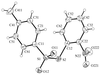
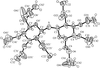
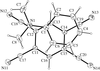
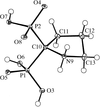
The four title CuII compounds, namely [Cu(fpy)Cl]ClO4, [Cu(etpa)Cl][CuCl4]1/2, [Cu(mopy)Cl]ClO4 and [Cu(pipy)Cl]PF6, where fpy is (2-furylmethyl)bis(2-pyridylmethyl)amine, etpa is 2-[bis(2-pyridylmethyl)amino]ethanolate, mopy is (2-morpholinoethyl)bis(2-pyridylmethyl)amine and pipy is (2-piperidinylethyl)bis(2-pyridylmethyl)amine, have potentially tetradentate tripodal ligands. In [Cu(fpy)Cl]ClO4 (shown opposite), the O atom of the furan moiety coordinates weakly to the Cu atom at a distance of 2.750 (3) Å.
Download citation


Download citation


The title compound crystallizes from light petroleum with two independent molecules in the asymmetric unit. In both molecules, one of the three benzyl ligands is η2-bonded to the Zr atom, indicated by the smallest Zr—CH2Ph angle and the consequently relatively short Zr⋯Cipso distance.
Download citation


Download citation


The structure of the cation in the title compound consists of the Cr atom bonded to the N atoms of the three 1,10-phenanthroline ligands, resulting in a distorted octahedral arrangement with the six Cr—N distances ranging from 2.040 (4) to 2.055 (4) Å. One of the perchlorate anions is disordered and is located around two special positions.
Download citation


Download citation


The title compound is a centrosymmetric dimer. The terephthalate moiety acts as a bridging ligand and the phenanthrolines as terminal ligands. The unique Cu atom is coordinated by two O and four N atoms in a distorted octahedral geometry, with Cu—O distances of 1.955 (2) and 2.815 (2) Å, and Cu—N distances of 2.008 (2)–2.216 (2) Å.
Download citation


Download citation


The isomorphous title compounds, (I>) and (II>), respectively, are monomeric and show distorted octahedral coordination with the monodentate dicyanamide ligands in cis positions. In both molecules, the metal–N(phenanthroline) bonds trans to the dicyanamide ligands are significantly longer than the other two.
Download citation


Download citation


The Ru atom in the title complex shows an octahedral coordination and a trans arrangement of the triphenylphosphine ligands. The observed bond lengths indicate a quinone rather than a diamide form for the o-benzoquinone diimine ligand. The complex molecule, in a special position, has crystallographic twofold symmetry.
Download citation


Download citation


The Ru atom in the title complex shows an octahedral coordination. The Cl− ion and the acetonitrile molecule are in the same plane as the o-benzoquinone diimine ligand, and the triphenylphosphine ligands are trans. Bond distances for the benzoquinone diimine ring establish a quinone arrangement.
Download citation


Download citation


The title compound consists of a hexaaquacobalt(II) cation and 4-toluenesulfonate anions, with no direct coordination of the anion to cobalt. The Co atom, in an octahedral geometry, is on a centre of inversion. The supramolecular structure consists of parallel layers of cations and anions. The anions are arranged with their sulfonate groups directed towards the cation layer in an alternating fashion and form hydrogen bonds with the water molecules of the cation.
Download citation


Download citation


The title compound is photochromic. Comparison with related complexes indicates that complexation with tricarbonylchromium has only minor effects on the structure of the chromene.
electronic papers (organic compounds)
Download citation


Download citation


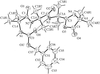
Our analysis of the title compound establishes its relative stereochemistry. The planar furan ring is twisted with respect to the steroid D ring and the crystal structure is stabilized by C—H⋯O hydrogen bonds and van der Waals interactions.
Download citation


Download citation


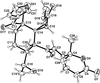
The X-ray structure determinations of the dihydrochloride monohydrate, (I>), and the trihydrochloride, (II>), of 7-methyl-3,7,11,17-tetraazabicyclo[11.3.1]heptadeca-1(17),13,15-triene are reported. Protonation occurs at the secondary amine N atoms in (I>) and at all the three amine N atoms in (II>) to which the Cl− ions are linked via N—H⋯Cl hydrogen bonds.
Download citation


Download citation


Sulfonium salts are known as photo acid generators (PAG) and have found a wide range of uses in photo-optical applications. Comparing the cations in the title compounds, (I>) and (II>), respectively, there is no significant effect of the methyl/phenyl substituents on the bonding characteristics of the central S atom. As expected, in both cations, the S—Ph bonds are somewhat shorter than the S—Me bonds and the interactions between cations and anions, in both crystal structures, are similar.
Download citation


Download citation


The crystal structure of the title compound in the monoclinic space group C2/c has a columnar packing arrangement with a 7.20 Å axis, a feature common to several tetraphenylmethanes.
Download citation


Download citation


In the two title adducts, (I>) and (II>), respectively, hydrogen bonds between the diammonium and carboxylate ions form a two-dimensional network parallel to the ab plane in (I>) and one-dimensional chains along the c axis in (II>). The cyclohexanediammonium ion in (II>) has a crystallographic twofold axis.
Download citation


Download citation


The title compound adopts a conformation with three cyanoethyl groups oriented in the same direction, suggesting the compound may behave as a potential tripodal ligand.
Download citation


Download citation


n-Butylammonium dihydrogen phosphate is ferroelastic. Ferroelastic switching is concomitant with the hopping of some H atoms involved in hydrogen bonds from donor to acceptor O atoms.
Download citation


Download citation


The title compound was found to exist as the endo–cis isomer with a pair of enantiomers in the asymmetric unit. The cyclohexene ring is folded about the methylene-to-CH(acetoxy) vector to give a boat conformation.
Download citation


Download citation


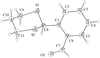
The title compound (BINAP oxide) was synthesized by direct oxidation of (R)-(+)-2,2′-bis(diphenylphosphino)-1,1′-binaphthyl (BINAP) with tert-butyl hydroperoxide in toluene solution. The angle between the naphthyl planes of the binaphthyl group is 94.17 (3)°.
Download citation


Download citation


The air-stable title compound was obtained by heating liquid acetylacetone with solid triphenylbismuth and was recrystallized from benzene. The product has two symmetry-equivalent but essentially identical molecules in the asymmetric unit, both showing an intramolecular O—H⋯O hydrogen bond.
Download citation


Download citation


In the molecules of the title compounds, (I>) and (II>), respectively, that of (I>) is twisted around the C—NO2 bond by 38.5 (1)°, while that of (II>) adopts a planar conformation stabilized by intramolecular hydrogen bonds, with a significant redistribution of π electrons.
Download citation


Download citation


The title compound is the product of the Diels–Alder reaction of anthracene with fumaric acid diethyl ester. The molecular C2 symmetry is nearly fulfilled in the crystal. Only the terminal torsion angles about the O—CH2 groups show significant differences.
Download citation


Download citation


Molecules of the title compound possess approximate non-crystallographic C2v symmetry, which is broken by the two chlorine substituents deviating from the ring plane. The closest intermolecular contacts are of 2.888 (2) Å between a carbonyl O atom and the C atom of a carbonyl group, forming chains of molecules.
Download citation


Download citation


In the crystal structure of the title compound, the pyridine ring makes a dihedral angle of 1.12 (9)° with the mean plane of the complete almost planar and crystallographically centrosymmetric molecule. There are stacks of parallel molecules along the a-axis direction, with alternate stacks having a herring-bone arrangement relative to each other and an interplanar spacing of 3.551 Å.
Download citation


Download citation


The molecule of the title compound is not planar. The dihedral angle between the two phenyl rings is 27.46 (7)° and in the dihydropyrazolopyrimidine ring the total puckering amplitude QT is 0.526 (3) Å. The structure is stabilized by both intra- and intermolecular C—H⋯O interactions, and by an intermolecular N—H⋯S hydrogen bond.


 journal menu
journal menu