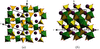issue contents
September 2004 issue

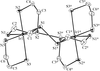
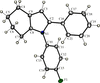
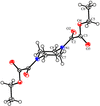
Cover illustration: A view of the unit cell of bis(tetraethylammonium) di-![[mu]](/logos/entities/mu_rmgif.gif) -chloro-bis[dichloropalladium(II)] at 292 K along the c axis. Displacement ellipsoids are drawn at the 50% probability level and H atoms are shown as small spheres of arbitrary radii. See Fábry, Dusek, Fejfarová, Krupková, Vanek & Nemec [Acta Cryst. (2004), C60, m426-m430].
-chloro-bis[dichloropalladium(II)] at 292 K along the c axis. Displacement ellipsoids are drawn at the 50% probability level and H atoms are shown as small spheres of arbitrary radii. See Fábry, Dusek, Fejfarová, Krupková, Vanek & Nemec [Acta Cryst. (2004), C60, m426-m430].
inorganic compounds
Download citation


Download citation


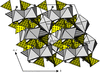
KSc(HAsO4)2 and RbScAs2O7 are both characterized by three-dimensional octahedral–tetrahedral frameworks, with intersecting tunnels hosting the alkali atoms. KSc(HAsO4)2 represents a new structure type, while RbScAs2O7 is the first reported diarsenate isotypic with KAlP2O7.
Download citation


Download citation


The triclinic (α) and monoclinic (β) modifications of CsSc(HAsO4)2 have three-dimensional octahedral–tetrahedral framework structures, with tunnels hosting the Cs atoms, and represent the first arsenate examples of their structure types. Strong to weak hydrogen bonds reinforce both frameworks.
Download citation


Download citation


The crystal structure of the new chromate Ce (CrO4)4·2H2O, prepared from an acidic aqueous solution at room temperature, is built from isolated CrO4 tetrahedra connected to isolated Ce(O,H2O)n (n = 8 and 9) polyhedra.
(CrO4)4·2H2O, prepared from an acidic aqueous solution at room temperature, is built from isolated CrO4 tetrahedra connected to isolated Ce(O,H2O)n (n = 8 and 9) polyhedra.
Download citation


Download citation


The peroskite oxynitride SrNbO2N crystallizes in space group 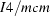 , both at room temperature and at 1.5 K. The Nb(O,N)6 octahedra are slightly rotated with respect to the tetragonal axis. A partially order of oxygen and nitrogen on the anionic sites was found.
, both at room temperature and at 1.5 K. The Nb(O,N)6 octahedra are slightly rotated with respect to the tetragonal axis. A partially order of oxygen and nitrogen on the anionic sites was found.
Download citation


Download citation


Hydrothermally synthesized Mg7(AsO4)2(HAsO4)4 has a framework structure built of edge- and corner-sharing MgO6, MgO4(OH)2, MgO5, AsO3(OH) and AsO4 polyhedra. The compound contains a very short hydrogen bond [O⋯O = 2.468 (3) Å].
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Open  access
access
 access
access Download citation


Download citation


Download citation


Download citation


Open  access
access
 access
access Download citation


Download citation


Open  access
access
 access
access Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation




 journal menu
journal menu