issue contents
August 2016 issue
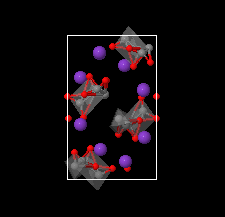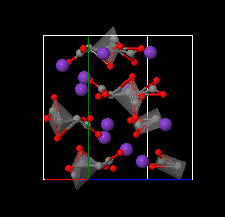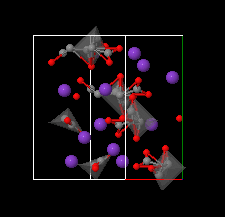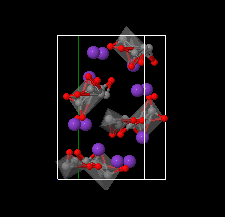
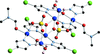
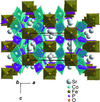
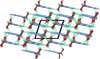
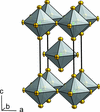
Cover illustration: The complex crystal structure of anhydrous potassium citrate, with a simple formula of [K3(C6H5O7)], was solved from powder diffraction data and optimized with the aid of density functional theory to reveal the citrate tri-anion in a trans,trans-conformation and a three-dimensional network of edge- and corner-sharing KO6 and KO8 polyhedra. See: Rammohan & Kaduk [Acta Cryst. (2016). E72, 1159-1162].
research communications
Download citation


Download citation


The crystal structure of the first trinuclear copper–pyrazolate (pz) complex with three different terminal ligands, including a strongly binding sulfate, is reported. The sulfate ion also acts as bridging ligand, leading to a dimeric structure, [Cu3(μ3-OH)(μ-4-Cl-pz)3(μ4-SO4)(DMF)(H2O)]2·4DMF·2H2O.
CCDC reference: 1489622
Download citation


Download citation


A linear geometry defined by a P,S-donor set is observed in the title polymorph; an intramolecular Au⋯O interaction is noted. The packing is consolidated by C—H⋯O and C—H⋯π interactions to generate a three-dimensional network.
CCDC reference: 1489737
Download citation


Download citation


Rubidium zinc iron(III) phosphate is isostructural to KCoAl(PO4)2. Its structure consists of a three-dimensional framework built up from corner-sharing PO4 and (Zn,Fe)O4 tetrahedra. This mode of linkage forms tunnels parallel to the [100], [010] and [001] directions in which the Rb+ ions are located.
CCDC reference: 1488120
Download citation


Download citation


The crystal structure of the title compound consists of discrete octahedral cobalt(II) complexes that are linked by a variety of hydrogen-bonding interactions into a three-dimensional network.
CCDC reference: 1491089
Download citation


Download citation


The absolute structure for the title compound, which has five chiral centres has been determined in this analysis. The supramolecular architecture comprises parallel zigzag chains formed through N—H⋯N and C—H⋯O hydrogen bonds, as well as intramolecular C—H⋯O, C—H⋯N and C—H⋯π interactions.
CCDC reference: 1490701
Download citation


Download citation


A distorted octahedral cis-N2S4 coordination geometry is found in the title solvated bimetallic complex. The packing features supramolecular layers sustained by O—H⋯O, O—H⋯N and N—H⋯O hydrogen bonding.
CCDC reference: 1489732
Download citation


Download citation


In the title copper(II) complex, the metal atom is coordinated by two N atoms and two O atoms from two bidentate (E)-1-[(2,4,6-tribromophenyl)diazenyl]naphthalen-2-ol ligands, forming a slightly distorted square-planar environment. In the isotypic nickel(II) and palladium(II) complexes, the metal atoms are located on centres of inversion, hence the metal coordination spheres have perfect square-planar geometries.
Download citation


Download citation


In the crystal a combination of N—H⋯O and asymmetric bifurcated O—H⋯(N,O) hydrogen bonds link the molecules into a three-dimensional network.
CCDC reference: 1491115
Download citation


Download citation


A new variety of tetrasodium copper(II) tris[molybdate(VI)] is characterized by the presence of infinite layers of composition (Cu1/Na1)2Mo3O14 parallel to the (100) plane, which are linked by MoO4 tetrahedra, forming a three-dimensional framework containing two types of hexagonal tunnels in which Na+ cations reside.
CCDC reference: 1487800
Download citation


Download citation


The crystal structures of three closely related benzothiazinone structures are reported. All three are conformationally similar having screw-boat puckering in the thiazine ring and C—H⋯π-type intermolecular interactions in the crystals.
Download citation


Download citation


In the title molecular salt 2-methyl-1H-imidazol-3-ium hydrogen oxalate dihydrate, N—H⋯(O,O) and O—H⋯O hydrogen bonds link the components into a bilayer-like assembly.
CCDC reference: 1491387
Download citation


Download citation


The crystal structure of the six-coordinate, highly ruffled, ferric porphyrinate bis(2-methylimidazole)[meso-tetrakis(p-tolyl)porphyrinato]iron(III) perchlorate is reported.
CCDC reference: 1471779
Download citation


Download citation


The 6-methyl-2-oxo-N-(quinolin-6-yl)-2H-chromene-3-carboxamide coumarin derivative displays intramolecular N—H⋯O and weak C—H⋯O hydrogen bonds, which probably contribute to the approximate planarity of the molecule [dihedral angle between the coumarin and quinoline ring systems = 6.08 (6)°]. The supramolecular structures feature C—H⋯O hydrogen bonds and π–π interactions, as confirmed by Hirshfeld surface analyses.
CCDC reference: 1491340
Download citation


Download citation


In both compounds, namely 2′-ferrocenyl-6′-methyl-6a'-nitro-6′,6a',6b',7′,9′,11a'-hexahydro-2H-spiro[acenaphthylene-1,11′-chromeno[3′,4′:3,4]pyrrolo[1,2-c]thiazol]-2-one, (I), and 6′-(4-methoxyphenyl)-6a'-nitro-6′,6a',6b',7′,9′,11a'-hexahydro-2H-spiro[acenaphthylene-1,11′-chromeno[3′,4′:3,4]pyrrolo[1,2-c]thiazol]-2-one, (II), an intramolecular C—H⋯O hydrogen bond forms an S(7) ring motif. In (I), molecules are linked via two different C—H⋯O hydrogen bonds, forming chains along [001] and [100]. In (II), they are linked through C—H⋯O hydrogen bonds, forming dimers with an 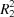 (10) ring motif while C—H⋯π interactions link the molecules in a head-to-tail fashion, forming chains along the a-axis direction.
(10) ring motif while C—H⋯π interactions link the molecules in a head-to-tail fashion, forming chains along the a-axis direction.
Download citation


Download citation


Two distinct coordination geometries, each based on a C2S4 donor set, are found in the title compounds, being based on an octahedron in (C6H5)2Sn(S2CN(Me)CH2CH2OMe)2 and a skew trapezoidal bipyramid in (C6H5)2Sn[S2CN(CH2CH2OMe)2]2.
Download citation


Download citation


A new layered tellurite has been synthesized, where the structural unit consists of [Cu2(TeO3)4]4− loop-branched chains of {Cu⋯Te⋯Cu⋯Te} squares, which are linked further into layers only through Y(O,H2O)8 polyhedra.
CCDC reference: 1493330
Download citation


Download citation


The transition metal orthophosphate, SrCo2Fe(PO4)3, crystallizes in an alluaudite-type structure. The chains characterizing the alluaudite structure are built up from edge-sharing [CoO6] octahedra linked together by PO4 tetrahedra.
CCDC reference: 1492743
Download citation


Download citation


In the crystal of the title purine derivative, molecules are linked by O—H⋯N, N—H⋯O and N—H⋯N hydrogen bonds, forming a three-dimensional framework.
CCDC reference: 1469976
Download citation


Download citation


Cycloaddition reaction between a menthone-based chiral nitrone and 2,5-dihydrofuran under microwave activation affords a enantiopure cycloadduct in good yields and with high stereoselectivity
CCDC reference: 1489217
Download citation


Download citation


Molecules of (2E)-1-(5-chlorothiophen-2-yl)-3-(2-methylphenyl)-prop-2-en-1-one (I) are linked into simple hydrogen-bonded C(5) chains, while three distinct conformers of (2E)-1-(anthracen-9-yl)-3-[4-(propan-2-yl)phenyl]prop-2-en-1-one (II) co-exist in the crystal but each conformer forms hydrogen-bonded C(8) chains, albeit of two different types.
Download citation


Download citation


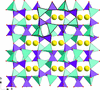
The crystal structure of anhydrous tripotassium citrate has been solved and refined using laboratory X-ray powder diffraction data, and optimized using density functional techniques.
Download citation


Download citation


The title compound was synthesized by treating (1S,3R,8S,9R,10S)-2,2-dichloro-3,7,7,10-tetramethyl-9,10-epoxytricyclo[6.4.0.01,3]dodecane with a concentrated solution of hydrobromic acid. It is built up from three fused rings: a cycloheptane ring, a cyclohexyl ring bearing alkene and hydroxy substituents, and a cyclopropane ring bearing two chlorine atoms.
CCDC reference: 1491732
Download citation


Download citation


The substitution of the aryl group at trans position with respect to the N atom in the imine part of 4,4-dihalogeno-1,1-aryl-2-azabuta-1,3-dienes [Ar2C=N—C(H)=CX2] by a CN group allows to get an almost perfectly planar molecule.
CCDC reference: 1491488
Download citation


Download citation


In the title 2-[(4,6-diaminopyrimidin-2-yl)sulfanyl]acetamides, both compounds have a folded conformation about the methylene C atom of the thioacetamide bridge, with the pyrimidine ring being inclined to the benzene ring by 56.18 (6) and 67.84 (6)°. In both molecules, there is an intramolecular N—H⋯N hydrogen bond stabilizing the folded conformation.
Download citation


Download citation


The structure of the title compound is similar to that of the related complexes [Co(C12H8N4S)2(N3)2] and [Ni(C12H8N4S)2(N3)2] in which the azide ion is substituted by the thiocyanate group. The CuN4S2 octahedron is more distorted than the CoN6 and NiN6 octahedra.
CCDC reference: 1494615
Download citation


Download citation


The title compound, glycidamide, was synthesized via the reaction of acrylonitrile and hydrogen peroxide. Both enantiomers occur as two crystallographically independent molecules in the asymmetric unit. In the crystal, molecules are linked by N—H⋯O hydrogen bonds forming β-sheet structures. The β-sheets are linked by weaker C—H⋯O hydrogen bonds, forming a supramolecular three-dimensional structure.
CCDC reference: 1490160
Download citation


Download citation


The asymmetric unit of the title polymeric compound contains one CuII atom, one coordinating water molecule, two 4-cyanobenzoate (CB) ligands and one coordinating N,N-diethylnicotinamide (DENA) molecule. The DENA ligands bridge adjacent Cu2+ ions, forming polymeric coordination chains running along the b axis.
CCDC reference: 1494903
Download citation


Download citation


The title compound, C11H6Cl2N2O2S, is a Schiff base that incorporates an N-bound 2,4-dichlorophenyl and a C-bound 5-nitrothiophene ring. The crystal structure features C—H⋯O hydrogen bonds and π–π stacking interactions. Geometric parameters from quantum-chemical calculations are in good agreement with experimental X-ray diffraction results.
CCDC reference: 1494850
Download citation


Download citation


In the title complex, the CrIII ion is coordinated to two methyliminodiacetate (mida) dianions acting as tridentate ligands through the N atom and two O atoms of each carboxylate group, in a distorted octahedral geometry. The partial ammonium cation is linked to two O atoms of carboxylate group from neighboring mida groups through N—H⋯O hydrogen-bonding interactions.
CCDC reference: 1494843
Download citation


Download citation


The title molecule, bergapten, a psoralen/furanocoumarin derivative, possesses photocarcinogenic and photomutagenic activity. In the crystal, molecules are linked via C—H⋯O hydrogen bonds, forming a three-dimensional framework.
CCDC reference: 1491854
Download citation


Download citation


In the title salt, the1-hexyl-N,N-dimethylpyridinium cations possess an extended chain conformation, while the bis(maleonitriledithiolato)nicklate(II) complex anion has a square-planar NiS4 geometry comprising four S-donor atoms from two bidentate chelate comprising maleonitriledithiolate ligands, with the Ni2+ cation lying on a crystallographic mirror plane. The crystal has a layered structure consisting of alternating cations and anions.
CCDC reference: 1491765
Download citation


Download citation


The structures of two facially coordinated Group VII metal complexes, fac-[ReCl(6,6′-dihydroxy-2,2′-bipyridine)(CO)3]·C4H8O and fac-[MnBr(6,6′-dihydroxy-2,2′-bipyridine)(CO)3]·C4H8O, are reported. These complexes are relevant to catalysis for CO2 reduction.
Download citation


Download citation


The crystal structures of two salts of unsymmetrical dimethyl hydrazine show different protonation patterns of the cation.
Download citation


Download citation


We present the first single-crystal X-ray structure analysis of NbF4 and compare some structural details with those obtained from previous powder X-ray diffraction studies. NbF4 crystallizes in the SnF4 structure type.
CCDC reference: 1496052
Download citation


Download citation


The title complexes are isotypic one-dimensional coordination polymers. The metal ions are bridged by binding to the N atoms of the two pyridine rings, and have an MN2Cl2 bisphenoidal coordination geometry. In the crystals of both compounds, the polymer chains are linked via pairs of C—H⋯Cl hydrogen bonds, forming corrugated slabs parallel to the ac plane.
Download citation


Download citation


The title compound, 5-[(4-carboxybenzyl)oxy]isophthalic acid (CIA), is non-planar with the two benzene rings being almost perpendicular to one another, making a 87.78 (7)°. In the crystal, molecules are linked by three pairs of O—H⋯O hydrogen bonds, forming undulating sheets parallel to the bc plane and enclosing 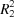 (8) ring motifs.
(8) ring motifs.
CCDC reference: 1494816
Download citation


Download citation


The title compound exists in the keto–amine tautomeric form. In the crystal, supramolecular layers are formed by O—H⋯O hydrogen bonding.
CCDC reference: 1496206


 journal menu
journal menu