issue contents
October 2020 issue
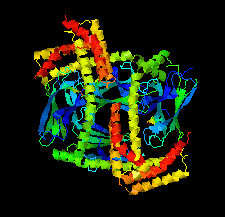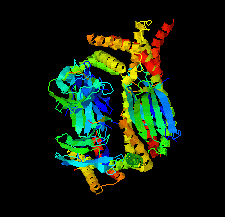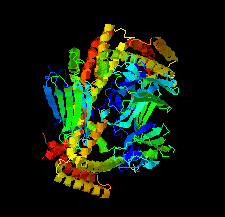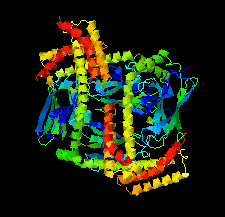
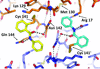
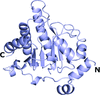
Cover illustration: Structure of the substrate-binding domain of heat-shock protein 70-x from the malaria parasite Plasmodium falciparum [Schmidt & Vakonakis (2020), Acta Cryst. F76, 495-500]. This structure supports the hypothesis that the parasite may partly exploit human chaperones for intra-erythrocytic trafficking and maintenance of its exported proteome.
research communications
 access
access access
access access
accessmethods communications
 access
access

 journal menu
journal menu













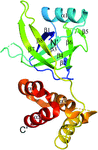
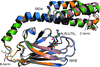


![[publBio]](/logos/publbio.gif)





