issue contents
November 2022 issue
editorial
CRYO | EM
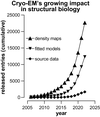
The 2022 meeting of the American Crystallographic Association in Portland was an inspiring event, addressing a range of both conventional and emerging themes in structural biology. The increasing emphasis at the conference on methods outside the conventional envelope of crystallography, especially cryo-electron microscopy, is discussed.
scientific commentaries
ELECTRON CRYSTALLOGRAPHY
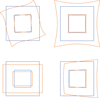
Electron diffraction patterns unavoidably contain in-plane distortions introduced by electromagnetic lens systems. Geometric correction of different distortion types allows the accuracy of lattice parameter determination to be improved in 3D electron diffraction data.
CRYO | EM
Commentary is given on a paper [Urzhumtsev & Lunin (2022). IUCrJ, 9, 728–734] proposing a new method for the analytic modelling of inhomogeneous resolution in electrostatic potential volumes and electron density maps for improved real-space refinement.
research letters
BIOLOGY | MEDICINE
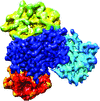
Family-wide structural analyses of human TRIM NHL domains reveal evolutionary divergence of their β-propeller architecture that might be essential for recruiting diverse interacting partners and for the roles of NHL domains as E3 ligases.
CRYO | EM
A decomposition of 3D oscillating functions results in analytic expressions for atomic model density maps distorted by inhomogeneous resolution and atomic positional disorder. Such decomposition extends the possibilities of real-space refinement of atomic models.
research papers
ELECTRON CRYSTALLOGRAPHY
Refinement of optical distortions in 3D electron diffraction data allows the determination of accurate lattice parameters and improves intensity integration.
BIOLOGY | MEDICINE
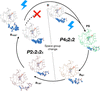
The use of room-temperature crystallography to probe protein dynamics on the second-to-minute timescale is demonstrated by monitoring the release of a photoequilibrium induced in crystals of a LOV domain. The slow protein dynamics components include the relaxation of a photoadduct and a crystalline phase transition. The result of the latter is the formation of a non-crystallographic dimer in which the C-termini of the two monomers reorder in distinct conformations on different timescales.
BIOLOGY | MEDICINE
Low-energy native-SAD phasing using a helium path could be used more routinely for solving challenging membrane protein structures.
PHYSICS | FELS
Using the CFEL TapeDrive, minimization of sample consumption and data collection times needed for serial synchrotron crystallography (SSX) were achieved and the first room-temperature structures of two enzymes are described as well as an assessment of the benefit of SSX for a radiation-sensitive protein.
PDB references: RT SSX structures:CTX-M-14, 7zpv; CTX-M-14 (10k dataset), 8af7; K. pneumoniae CTX-M-14 (5k dataset), 8af8; N. haematococca GH11 xylanase (40k dataset), 8af4; GH11 xylanase (10k dataset), 8af5; GH11 xylanase (4k dataset), 8af6; GH11 xylanase (1k dataset), 7zq0; cofactor-free A. flavus urate oxidase complex, 7qar


 journal menu
journal menu





 access
access