issue contents
ISDSB 2019
Edited by Atsushi Nakagawa, John Helliwell and Yuriko Yamagata
This virtual issue collects together articles from the 6th International Symposium on Diffraction Structural Biology (ISDSB) 2019.
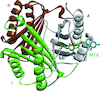
Cover illustration: Professor Nobuhisa Watanabe, who died from leukemia on 26 March 2019, and his work on high-pressure protein crystallography of hen egg-white lysozyme [Yamada et al. (2015). Acta Cryst. D71, 742-753]. The opening session of ISDSB2019 was dedicated to Professor Watanabe, and his scientific achievements in protein crystallography and beamline instrumentation were highlighted in the meeting.
Free 

Introducing the special issue on diffraction structural biology available at https://journals.iucr.org/special_issues/2021/ISDSB2019.
Open  access
access
 access
accessThe three probes of the structure of matter (X-rays, neutrons and electrons) in biology have complementary properties and strengths. For the study of combined states of order and disorder, NMR crystallography is also applicable. Overall, to understand a biological system, the requirements are physiologically relevant results. Reporting of protein crystal structures, and their associated database entries, could usefully indicate how close to the biological situation they are.
Open  access
access
 access
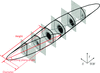
accessKey structural biology experiments that have sought to elucidate how potassium ions permeate and pass through the selectivity filter of potassium ion channels are reviewed.
Open  access
access
 access
accessThe current capabilities of and future upgrade plans for the beamlines supporting structural biology at the Canadian Light Source are described.
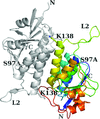
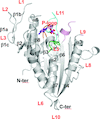
Based on the structure of the N-terminal region of the S97A mutant of the stomatin operon partner protein PH1510 (1510-N), it was found that the dimeric form differs greatly from those determined previously. This result indicates that 1510-N can greatly change the form of its dimer.
PDB reference: S97A mutant of 1510-N, 6m4b
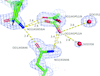
Based on crystal structures of HIRAN in complex with duplex DNA, the mechanism of replication-fork regression underlying the sequence-independent recognition of nucleobases by HIRAN is discussed.
Open  access
access
 access
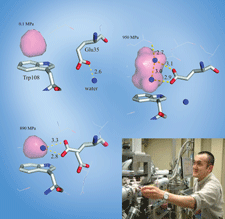
accessThe complex of lysozyme with an N-acetylglucosamine tetramer shows a relatively strong hydrogen-bond network around a catalytic residue via high-resolution X-ray structural analysis. This indicates a potentially different hydrolysis mechanism to that through a glycosyl intermediate, and this is expected to be proved using neutron experiments.
Open  access
access
 access
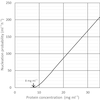
accessA semi-empirical model to estimate the optimum conditions for the growth of large protein crystals has been developed.
Crystal structures of the glycosyltransferase PaGT3 crystallized in the presence of crown ethers provide a basis for understanding its acceptor-recognition mechanism.
Engrailed homeodomain (EHD) is a low-molecular-weight DNA-binding protein that can potentially act as a molecular tool to target desired DNA sites in cells. The crystal structure of tandemly connected EHDs with R53A mutations revealed that the mutation does not interfere with the important base-specific interactions of the wild-type protein; instead, it works to properly modulate the DNA-binding affinity, leading to precise recognition of the target site.
PDB reference: (EHD[R53A])2–DNA complex, 6m3d
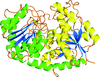
The crystal structure of DcsB, an Nω-hydroxy-L-arginine hydrolase found in the D-cycloserine biosynthetic pathway, is reported.
Large crystals for neutron diffraction experiments were cooled anaerobically by two methods, cooling in a cold gas stream and plunge-cooling into liquid nitrogen, and their respective effects on crystal quality were compared. Gas-stream cooling was sufficient to suppress the increase in mosaicity over the entire crystal, leading to success in the first data collection for a membrane-bound hydrogenase from Desulfovibrio vulgaris in its active reduced form.
A large single crystal of the Y116S variant of transthyretin was grown and neutron diffraction data were collected to 1.9 Å resolution using the IBARAKI biological crystal diffractometer (iBIX) at the Japan Proton Accelerator Research Complex (J-PARC).
Open  access
access
 access
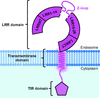
accessThe innate immune receptor TLR8 can be positively or negatively regulated by small chemical ligands. Structural views of agonist-bound and antagonist-bound forms have revealed the mechanisms underlying agonism and antagonism.
Open  access
access
 access
accessA new room-temperature (RT) data-collection method for microcrystals was developed by combining serial synchrotron rotation crystallography with the humid air and glue-coating method. The RT data-collection strategy for micro-crystallography was evaluated by examining the efficiency, the influence of non-isomorphism and radiation damage, and the effectiveness of increasing the number of merged images. An equation was proposed that relates the achievable resolution to the total photon flux used to obtain a data set.
Open  access
access
 access
accessCrystallization and structure determination of the motor domain of centromere-associated protein E in complex with its inhibitor was performed. In the determined structure, endogenous ADP was observed in the nucleotide-binding site instead of the inhibitor.
PDB reference: CENP-E motor domain, 6m4i
X-ray crystal structures were determined of the wild type and an inactive mutant of α-amylase from the earthworm Eisenia fetida. Structural analyses reveal the molecular properties that are responsible for catalytic activity at low temperatures and substrate recognition.
Structure determination of the complex of macrophage migration inhibitory factor (MIF) with methotrexate (MTX) and fragment molecular-orbital calculations quantified the interaction between MTX and MIF and revealed the amino acids that are effective in the interaction of MTX and MIF.
PDB reference: MIF–MTX complex, 5xej
A crystal-mounting platform has been developed for native SAD phasing. This platform includes a newly designed solution-free tool and an automatic freezing–extracting robot for use at synchrotron facilities.
Open  access
access
 access
accessAn improved, fully automated protein crystallization and monitoring system, PXS2, can perform crystallization using smaller protein samples than the previous version. In addition, PXS2 can be used for membrane-protein crystallization using the bicelle and LCP methods.
Open  access
access
 access
accessCryocrystallography, which is commonly used in macromolecular crystallography, may sometimes reduce the quality of diffraction data and the visibility of crystals owing to frost adhesion. A device has been developed to remove frost by drizzling liquid nitrogen over the crystals, which enabled noise reduction of diffraction images and the centering of crystals with low visibility owing to frost adhesion.


 menu
menu