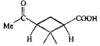
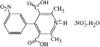
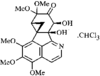
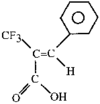
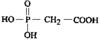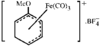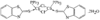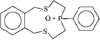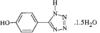issue contents
January 1997 issue
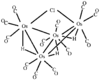
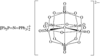
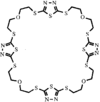
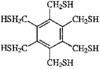
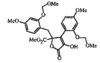
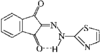
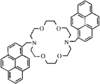
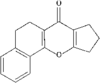
Cover illustration: A Tetrameric Macrocycle with 2,5-Dithio-1,3,4-thiadiazole Subunits, see Rugutt, Fronczek, Watkins & Pappalardo, pages 81-84. Displacement ellipsoids are plotted at the 75% probability level. S atoms are shown in yellow, N atoms are shown in blue, O atoms are shown in red and C atoms are shown in green.
editorial
Free 

inorganic compounds
Download citation


Download citation


The structure of K3Al2As1.92P1.08O12, prepared by solid-state reaction, contains AlO4 and (As,P)O4 tetrahedra which share corners to form a three-dimensional framework. The structure is related to those of the compounds MIMIIXO4 and tridymite.
Download citation


Download citation


The structure of NaInAs2O7, prepared by solid-state reaction, contains InO6 octahedra and As2O7 diarsenate groups. These are linked by In—O—As bridges to form a three-dimensional framework containing cages within which the Na+ ions are located.
Download citation


Download citation


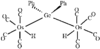
The structure of (NH4)4(H6ZnMo6O24).6H2O is composed of (H6ZnMo6O24)4− anions, NH4+ cations and water molecules linked by hydrogen bonds. The anion is formed from six edge-sharing octahedral MoO6 units surrounding a central ZnO6 octahedron with all metal ions lying in a common plane.
Download citation


Download citation


Os3Sn15O14 is a metal-rich stannate containing Os-centered Sn6 octahedra. The OsSn6 units are linked to form triple chains in which the oxidation state of the linking Sn atoms is +1 and the oxidation state of the terminal Sn atoms is +2.
Download citation


Download citation


K2MgWO2(PO4)2 undergoes at least five phase transitions between room temperature and its melting point. The structure of the room-temperature form is triclinic pseudo-tetragonal (space group P1) and is composed of chains of alternately corner-linked [WO6] and [MgO6] octahedra connected via [PO4] tetrahedra.
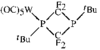
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


international union of crystallography
Free 



 journal menu
journal menu