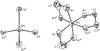issue contents
September 2000 issue
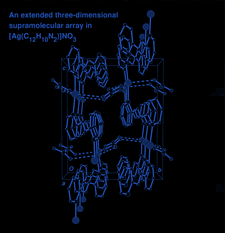
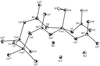
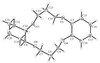
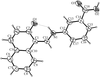
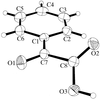
Cover illustration: A perspective view of the molecular packing in catena-poly[[silver(I)-![[mu]](/logos/entities/mu_rmgif.gif) -[(E)-1,2-bis(2-pyridyl)ethylene-N:N']] nitrate]. See Tong & Chen [Acta Cryst. (2000), C56, 1075-1076].
-[(E)-1,2-bis(2-pyridyl)ethylene-N:N']] nitrate]. See Tong & Chen [Acta Cryst. (2000), C56, 1075-1076].
inorganic compounds
Download citation


Download citation


Sr3Ga3(BO3)4O(OH) represents a new ortho-borate structure type containing the uncommon square-pyramidal GaO4OH unit.
Download citation


Download citation


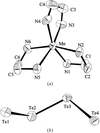
The title compound is composed of one-dimensional [Ti3Te113−] chains built by face-sharing pentagonal TiTe7 bipyramids and distorted TiTe6 octahedra. These chains adopt hexagonal closest packing along the [101] direction. Rb atoms are located among these chains.
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


The structure of the title compound, obtained solvothermally at 473 K, has been determined by single-crystal X-ray diffraction. It consists of one-dimensional [AlP2O8H]2− macroanions, connected to each other by pentanediammonium cations.
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


A new three-dimensional open-framework germanate was hydrothermally synthesized and its structure determined by single-crystal X-ray diffraction. The framework is built of [Ge9O22(OH)4] units and contains intersecting channels in three directions formed by an eight- and two ten-membered rings.
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


addenda and errata
Download citation


Download citation


Free 

A full description for the structure of bis(O-isopropyldithiocarbonato)cadmium(II) is presented
electronic papers (inorganic compounds)
Download citation


Download citation


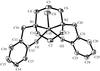
The new caesium tantalum chalcogenide Cs6Ta4S22 was prepared from the reaction of Cs2S3, Ta and S. The compound is isotypic with Rb6Ta4S22 and A6Nb4S22 (A = Rb, Cs), and consists of discrete [Ta4S22]6− anions and Cs+ cations.
electronic papers (metal-organic compounds)
Download citation


Download citation


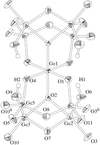
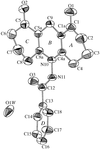
The title binuclear nickel(II) complex has a slightly distorted octahedral coordination. The pyridazine and two methylpyridine rings of the bridging tetradentate ligand are coplanar to within 0.181 (3) Å.
Download citation


Download citation


In the title compound, the Ni atom is bonded centrosymmetrically by four N and two O atoms in a distorted octahedral coordination. The acetate ligands form intermolecular hydrogen bonds to imidazole moieties, giving rise to a two-dimensional layer structure.
Download citation


Download citation


The Cu atom in the title complex has an N4O2 octahedral coordination geometry, the Cu atoms being bridged by the bipyridine and chelated by the phenanthroline heterocycles. Adjacent molecules are linked into a zigzag chain running along the c axis of the monoclinic unit cell. The chains are connected through lattice water molecules to produce a layer structure.
Download citation


Download citation


In the title complex, the Zn atom is located at a center of symmetry and has a distorted octahedral geometry, with Zn—O distances in the range 2.052 (2)–2.181 (2) Å. Hydrogen bonds involving the water molecules make up a three-dimensional network.
Download citation


Download citation


In the title crystals, (IIIa)> and (IIIb)>, respectively, the bonding parameters differ significantly. That also applies for the known structure of the corresponding dichloromethane solvate. Complex (IIIb)> has a crystallographic centre of symmetry.
Download citation


Download citation


The title compound crystallizes as a linear polymeric compound with one pyrimidine ligand bridging between two CuII atoms and a second pyrimidine ligand coordinated in a monodentate manner. The coordination geometry around the CuII atom is a distorted octahedron including a long Cu⋯O bond.
Download citation


Download citation


The title compound is a monomeric trans-octahedral complex, with the SnIV atom at the centre of symmetry, planar 2-mercaptopyridine zwitterions and SnS2C2 groups in twist–envelope conformations. The 2-mercaptopyridine ligands are monodentate.
Download citation


Download citation


The structure of the title compound was redetermined and the correct C2/c space group was assumed instead of the previously reported P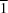 space group. The two independent molecules reported for the triclinic cell are actually related by a twofold axis.
space group. The two independent molecules reported for the triclinic cell are actually related by a twofold axis.
Download citation


Download citation


The structures of the two title compounds consist of discrete [CuCl4]2− anions with two monoprotonated C2N3H8+ cations for (I>) and a diprotonated C8N4H142+ cation for (II>). The [CuCl4]2− anions have flattened tetrahedral geometries.
Download citation


Download citation


The title complex is the first crystal structure reported with dimethyl sulfoxide coordinated to CoIII. It crystallizes with two independent molecules in the asymmetric unit. A localized non-crystallographic inversion centre is observed between the two cations.
Download citation


Download citation


The imino ligand displays weak π-acceptor capabilities and, in contrast to Fischer-type carbene complexes, is not positioned on the bisecting line of the angle between two CO ligands.
Download citation


Download citation


The cation of the title complex consists of an Ru atom in a pseudo-octahedral coordination environment, with two chelating 1,2-bis(diphenylphosphino)ethane (dppe) ligands and one chelating acetate ligand. The hexafluorophosphate anion and methanol solvate molecules are unexceptional.
electronic papers (organic compounds)
Download citation


Download citation


The fused six- and five-membered rings adopt chair and twist conformations, respectively, in the title catanospermine derivative.
Download citation


Download citation


In the title compound, the tropone ring is at an angle of 50.3 (1)° to the crown ether. The conformation of the ethereal moiety is S(g)C(g)C(t)O(t)C(g)C(t)O(t)C(g)C(t)O(t)C(g)C(g)S (t = trans, g = gauche). The CH2—O bonds are all trans, which is different to the case in the Hg complex of the title compound.
Download citation


Download citation


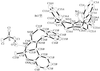
The absolute configuration was determined for the title compound. The cation contains a 14-membered macrocyclic lactone and two sugars, namely cladinose and desosamine. The six-membered rings of the sugars adopt chair conformations.
Download citation


Download citation


In the title compound, the dihedral angle between the two phenyl rings is 15.69 (6)°. An intermolecular hydrogen-bonding association exists between the hydroxy group and one of the azo N atoms.
Download citation


Download citation


In the title compound, the naphthyl unit is planar and the benzopyran unit is nearly planar. These two moieties are inclined at an angle of 9.10 (6)° with respect to one another.
Download citation


Download citation


In the title compound, there are two independent molecules in the asymmetric unit having essentially the same geometry, with central O—N—C—H torsion angles of −177.7 (1) and −178.4 (1)°, and with dihedral angles between the phenyl rings of 125.8 (6) and 126.1 (6)°.
Download citation


Download citation


The molecular structure of the title compound, which is an intermediate in the synthesis of trifluoromethyl-substituted naphthalenes, adopts a bent conformation. Intramolecular O—H⋯F and intermolecular O—H⋯O interactions form a bifurcated hydrogen bond which produces a supramolecular assembly of head-to-tail dimers.
Download citation


Download citation


In the title compound, the N atom of one molecule is linked to the amido-O atom of an adjacent molecule across a center of symmetry by a cyclic hydrogen bond [N⋯O = 2.888 (4) Å]. The molecule is planar and all bond distances and angles are normal, except for the elongated C(=O)—C(=O) single bond of 1.554 (4) Å.
Download citation


Download citation


The three title compounds, i.e. (I)–(III), respectively, are almost planar and show quite strong conjugation between the benzoxazine and nitroaryl moieties, with the exception of the carbonyl groups, which deviate from the planes of the benzoxazine rings and cause significant deviation of the bond angles around the carbonyl C atoms from 120°.
Download citation


Download citation


In the title compound, the C—C—C—C torsion angle of the molecular skelton is −52.7 (4)° and the tertiary H atoms attached to the central C atoms are in a trans arrangement. The hydroxyl groups form hydrogen bonds with the carbonyl O atoms of neighbouring molecules resulting in a three-dimensional framework.
Download citation


Download citation


In the title compounds, (I>) and (II>), respectively, the amide groups are rotated out of the central C—C—C plane by ca 76° in (I>) and by 70–73° in (II>). Compound (I>) has crystallographic mirror symmetry perpendicular to the molecular plane. There are N—H⋯O intermolecular hydrogen bonds in (I), and N—H⋯O and C—H⋯O intermolecular hydrogen bonds in (II), and each amide O atom acts as an acceptor in two hydrogen bonds.
Download citation


Download citation


In the title compound, determined in order to obtain coordinates for molecular-orbital calculations, intermolecular N—H⋯O and C—H⋯O hydrogen bonds link the molecules into continuous two-dimensional sheets parallel to the (100) plane.
Download citation


Download citation


The structure of a new triclinic modification of 2′,6′-dimethoxyflavone is described. In total, it contains Z = 6 molecules of three geometries characterized by different torsion angles of the substituted phenyl ring.
Download citation


Download citation


The title compound is a steroid with a dienone structure and six stereogenic centers. The configuration of the 11 and 13 centres is R. These steroids are substrates for estradiol derivatives, bearing an alkyl chain in the 7α position.
Download citation


Download citation


The crystal structure of L-kynurenine, a well known tryptophan metabolite, has been determined as the sulfate. The molecule has a trans zigzag carbon-skeletal conformation and the two amino groups are protonated. The two amino and the carboxyl groups are located on the same side of the molecule. The crystal structure is stabilized by intermolecular hydrogen bonds involving the water molecules and the sulfate ion.
Download citation


Download citation


The title compound is the initially formed product from acetylation of 9-fluorenyllithium. Its structure is the sp rotamer whose acetyl group is not rotationally restricted but whose C—C=O plane is essentially perpendicular to the fluorene ring, in contrast to 9-pivaloylfluorene, which is rotationally restricted and exists exclusively as its ap rotamer.
Download citation


Download citation


The title compound shows no unusual features. The structure contains two molecules of water of crystallization.


 journal menu
journal menu