issue contents
January 2006 issue
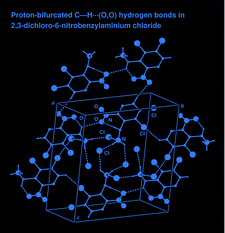
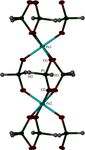
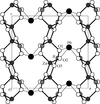
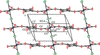
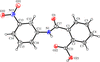
Cover illustration: The intermolecular interactions in 2,3-dichloro-6-nitrobenzylaminium chloride, including the three-center hydrogen-bond configuration. For clarity, only relevant H atoms and intramolecular interactions are shown. See Rathore, Narasimhamurthy, Vijay, Yathirajan & Nagaraja [Acta Cryst. (2006), C62, o36-o38].
inorganic compounds
Download citation


Download citation


The title compound, an Na–Mn–Al-bearing phosphate, has been synthesized by the hydrothermal technique. The crystal structure is similar to that of the natural mineral alluaudite.
Download citation


Download citation


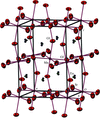
Eu0.6Sr0.4MnO3 crystallizes with an orthorhombic (Pbnm) perovskite-type structure and displays split positions of the Eu3+ and Sr2+ cations.
Download citation


Download citation


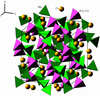
Tricesium lithium dicobalt tetrakis(tetraoxomolybdate), Cs3LiCo2(MoO4)4, and trirubidium lithium dizinc tetrakis(tetraoxomolybdate), Rb3LiZn2(MoO4)4, are isotypic with Cs6Zn5(MoO4)8 and contain frameworks of MoO4 and (M2/3Li1/3)O4 tetrahedra (M = Co and Zn) with alkaline cations in cuboctahedral cavities. The frameworks are close to that of 12CaO·7Al2O3 and the cations repeat the Y3Au3Sb4 structure.
Download citation


Download citation


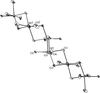
The structure of Fe(H2PO2)3 consists of [001] chains of octahedral Fe3+ cations (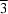 symmetry) bridged by bidentate hypophosphite anions.
symmetry) bridged by bidentate hypophosphite anions.
Download citation


Download citation


PbZn2(BO3)2 crystallizes in the space group Pccn, with the Pb atom at a site with imposed twofold symmetry. The compound represents a new structure type in which ZnBO3 layers are bridged by Pb2+ cations, giving rise to a three-dimensional framework.
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Open  access
access
 access
access Download citation


Download citation


Open  access
access
 access
access Download citation


Download citation


Open  access
access
 access
access Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Open  access
access
 access
access Download citation


Download citation


Open  access
access
 access
access Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Open  access
access
 access
access Download citation


Download citation


Open  access
access
 access
access Download citation


Download citation


Open  access
access
 access
access Download citation


Download citation


Open  access
access
 access
access Download citation


Download citation




 journal menu
journal menu