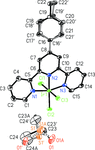
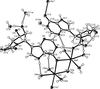
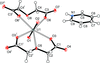
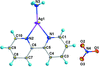
issue contents
March 2010 issue
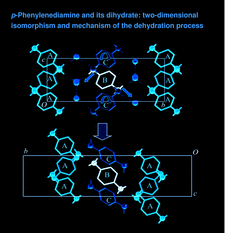
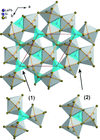
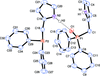
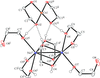
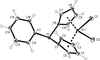
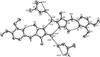
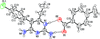
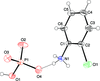
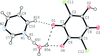
Cover illustration: The proposed mechanism for the transformation of p-phenylenediamine dihydrate into p-phenylenediamine. One set of arrows show the direction of rotation of the C molecules, and the other set indicates pairs of water molecules replaced by the amino groups of the B molecules. See Czapik, Konowalska & Gdaniec [Acta Cryst. (2010), C66, o128-o132].
inorganic compounds












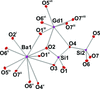
metal-organic compounds


















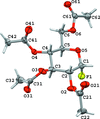
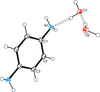
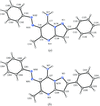
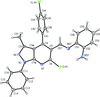
organic compounds






















addenda and errata





 journal menu
journal menu