issue contents
May 2022 issue
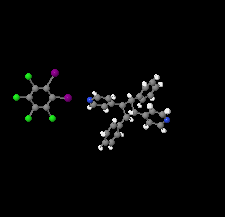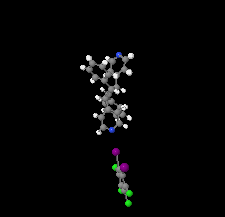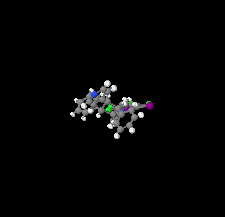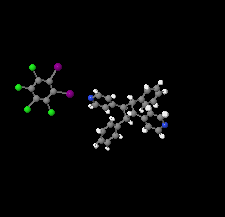
Cover illustration: Supramolecular networks can be designed using self-assembly of small molecules for example by halogen bonding. This report describes the use of I⋯N halogen bonds between 1,2-diiodoperchlorobenzene and the photoproduct rctt-1,3-bis(pyridine-4-yl)-2,4-diphenylcyclobutane resulting in zigzag chains due to the ortho-position of the two involved I atoms. Neighbouring chains generate two-dimensional sheets by Cl⋯π interactions with pyridyl and phenyl rings. See: Dunning, Bosch & Groeneman [Acta Cryst. (2022). E78, 506–509].
research communications
Download citation


Download citation


The crystal structure of the title compound comprises two different CoII complexes, one mononuclear and the other dinuclear, with the enantiopure, bidentate (−)-5,6-pinenebipyridine ligand. All three coordination polyhedra around the CoII cations are distorted trigonal bipyramids.
CCDC reference: 2163153
Download citation


Download citation


The crystal structure of methanol solvate of berberine chloride, 9,10-dimethoxy-5,6-dihydro-2H-7λ5-[1,3]dioxolo[4,5-g]isoquinolino[3,2-a]isoquinolin-7-ylium chloride methanol monosolvate, was determined and its Hirshfeld surface analysis was performed.
CCDC reference: 2161706
Download citation


Download citation


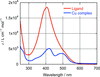
A copper(II) complex with the (E)-2-methyl-4-(quinolin-8-yldiazenyl)benzene-1,3-diol ligand was prepared and structurally characterized. The UV–Vis absorption spectra of the ligand and the complex are reported.
CCDC reference: 2162331
Download citation


Download citation


In the title compound, the molecule adopts an anti-configuration, in which two partially overlapped carbazole fragments form an intramolecular slipped π–π interaction. In the crystal, the molecules are cross-linked via intermolecular C—H⋯N hydrogen bonds and C—H⋯π interactions into a three-dimensional network.
CCDC reference: 2161895
Download citation


Download citation


The title polyoxometalate salt, (NH4)2[Zn(DMF)(H2O)5]2[V10O28]·4H2O, comprising a cationic Zn2+ complex, belongs to a group of relatively rare decavanadates with complex counter-ions, previously including only eight crystal structures with zinc(II) components. In the crystal structure, the ions are linked by strong O—H⋯O hydrogen bonds between the coordinated water ligands and the decavanadate, with the O⋯O distances ranging from 2.660 (2) to 2.893 (2) Å.
CCDC reference: 2156016
Download citation


Download citation


The X-ray crystal structure, and Hirshfeld surface analysis of a sulfonyl urea analogue, 4-bromo-N-(propylcarbamoyl)benzenesulfonamide is reported.
CCDC reference: 2164397
Download citation


Download citation


In the crystal structure of the title compound, [Zn(cyclam)]2+ cations and SbS43– anions are present, which are linked to acetonitrile and water solvate molecules via intermolecular hydrogen bonding.
CCDC reference: 2164599
Download citation


Download citation


The ability of 4-[(benzylamino)carbonyl]-1-methylpyridinium bromide salt to form hydrates was studied. Hirshfeld surfaces analysis was performed for identification of intermolecular interactions.
CCDC reference: 2164796
Download citation


Download citation


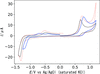
Nickel(II) Schiff base complexes containing thiolate S and polyamine N donor atoms exhibit electrocatalytic activity for proton reduction. The piperazine moiety in the Schiff base ligand gives a smaller bite angle, which is effective in reducing the overpotential.
Download citation


Download citation


The formation of a halogen-bonded zigzag molecular network based upon 1,2-diiodoperchlorobenzene and the head-to-tail photoproduct rctt −1,3-bis(pyridin-4-yl)-2,4-diphenylcyclobutane is reported. The co-crystal is sustained by I⋯N halogen bonds where the photoproduct acts as a linear linker while the donor behaves as a bent two-connected node within the zigzag chain.
CCDC reference: 2133162
Download citation


Download citation


Six piperazinium salts are reported, five of them are hydrated and two crystallized as 2:2 salts. They exhibit asymmetric units of a common 4-nitrophenylpiperazine cation and different p-substituent benzoate anions. Their crystal structures mainly pack as chains stabilized by strong N—H⋯O and O—H⋯O hydrogen bonds and other weak interactions such as C—H⋯O and C—H⋯π.
Download citation


Download citation


The title complexes feature distorted trigonal–planar CuS2X (X = Br, I) coordination geometries for the metal ions. The presence of the acetone solvent molecule in the iodide complex plays an important role in the packing.
Download citation


Download citation


In the crystal, the two independent molecules are linked through the water molecules by intermolecular O—H⋯O and O—H⋯N hydrogen bonds, producing chains along the b-axis direction. These chains are linked with neighboring chains parallel to the (103) plane via C—H interactions, generating ribbons along the b axis. The van der Waals interactions between the strips ensure the stability of molecular packaging.
CCDC reference: 2168161
Download citation


Download citation


C—H⋯O and C—H⋯F hydrogen bonds link molecules in the crystal into layers parallel to (011). The crystal packing is consolidated through C—Br⋯π and C—F⋯π interactions, as well as π–π stacking interactions.
CCDC reference: 2168678
Download citation


Download citation


The crystal structure of a new one-dimensional [EuIII(phth)(OAc)(H2O)] coordination polymer and its room-temperature photoluminescent properties are reported.
CCDC reference: 2168116

 journal menu
journal menu