issue contents
July 2019 issue
editorial
MATERIALS | COMPUTATION
This Editorial discusses recent development in computational and materials structural science as exemplified by recent articles published in IUCrJ.
scientific commentaries
BIOLOGY | MEDICINE
A practical approach for obtaining S-SAD data from native protein microcrystals with low-wavelength synchrotron radiation [Guo et al. (2019), IUCrJ, 6, 532–542] is presented in this issue of IUCrJ.
CHEMISTRY | CRYSTENG
Khandavilli et al. [(2019), IUCrJ, 6, 630–634] show the superior plasticity in hydrates of the pharmaceutical drugs, pregabalin and gabapetin, compared with their anhydrous forms. The water in the structure is believed to act as a lubricating agent in the packing of hydrates, thus facilitating slippage of molecules in the plastic bending of the crystals under external mechanical stress.
feature articles
NEUTRON | SYNCHROTRON
Resonant and non-resonant X-ray magnetic diffraction under high pressure (above 40 GPa) is comprehensively discussed and reviewed.
research letters
BIOLOGY | MEDICINE
Medical contrast media are presented as possible tools to probe the internal structure of soft-matter and biological systems by small-angle X-ray scattering solvent contrast variation.
CRYO | EM
A pipeline tool called Namdinator is presented that enables the user to run a molecular dynamics flexible fitting (MDFF) simulation in a fully automated manner, online or locally. This provides a fast and easy way to create suitable initial models for both cryo-EM and crystallography, and help fix errors in the final steps of model building.
research papers
BIOLOGY | MEDICINE
Low-energy native-SAD phasing from microcrystals of less than 10 micrometres in size is demonstrated at a synchrotron microdiffraction beamline.
PDB reference: thaumatin, 6o8a
BIOLOGY | MEDICINE
A method is described that enables both time- and sample-efficient X-ray crystallographic data collection from radiation-sensitive protein microcrystals and direct comparison of the resulting dose-resolved serial synchrotron and damage-free X-ray free-electron laser structures. It is demonstrated that real-space extrapolation from a synchrotron dose series reproduces key features of the damage-free structure obtained using an X-ray free-electron laser.
PDB references: Dye-type peroxidase Aa from Streptomyces lividans, MSS 78.4 kGy structure, 6i8p; 274.4 kGy structure, 6q3e; 235.2 kGy structure, 6q3d; 164 kGy structure, 6i8k; 98.4 kGy structure, 6i8i; 32.8 kGy structure, 6i7z; 39.2 kGy structure, 6i8o; 196 kGy structure, 6q34; 117.6 kGy structure, 6i8q; 65.6 kGy structure, 6i8e; 156.8 kGy structure, 6q31; 131.2 kGy structure, 6i8j; 313.6 kGy structure, 6ibn; SFX structure of damage-free ferric state of dye-type peroxidase Aa from Streptomyces lividans, 6i43
CHEMISTRY | CRYSTENG
A Heusler Cr3Al thin film with a perfect DO3 structure is successfully prepared for the first time and its physical properties are investigated.
CHEMISTRY | CRYSTENG
Download citation


Download citation


Combinations of ab initio calculations and modelling against X-ray diffraction data are used to derive the free energy of polymorphic crystals. The results are discussed in relation to the relative stability and phase transitions of the enantiotropically related polymorphs.
BIOLOGY | MEDICINE
Glucosylglycerate accumulates to high levels in nitrogen-starved mycobacteria. Here, a thorough structural and biochemical characterization unveils the molecular determinants of the substrate specificity and catalytic mechanism of a mycobacterial glucosylglycerate hydrolase, a highly conserved enzyme involved in the fast recovery of rapidly growing mycobacteria from nitrogen starvation.
NEUTRON | SYNCHROTRON
Equations and methodology for correcting the dominant systematic in fluorescence measurements are provided. They are particularly appropriate for X-ray absorption spectroscopy in fluorescence and for multi-pixel detection.
MATERIALS | COMPUTATION
The shapes of the HOMOs/LUMOs and the relative positions of adjacent molecules decide the size of the intermolecular electronic couplings.
MATERIALS | COMPUTATION
First-principles calculations suggest that C1b-ordered FeMnZ (Z = Al, Ga, In and Al0.5Ga0.5) alloys are half-metallic fully compensated ferrimagnets with a spin-gapless semiconducting nature. In such materials, full spin polarization, spin-gapless properties and absence of external stray fields, thus exhibiting minimal energy losses, are expected.
BIOLOGY | MEDICINE
Martensitic transformation in crystals of reversibly switchable fluorescent protein Tetdron can be photo-induced by illumination with 400 nm light at room temperature. The phase transition results in tetramerization of the protein in crystallo coupled with chromophore deprotonation as demonstrated by X-ray photocrystallography and spectroscopic measurements.
CHEMISTRY | CRYSTENG
Download citation


Download citation


The hydrate forms of the zwitterionic drugs Pregabalin and Gabapentin show plastic bending in a single crystal, whereas the anhydrous equivalents are brittle. A close structural analysis attempts to justify such behaviour.
NEUTRON | SYNCHROTRON
Angular cross-correlation analysis of X-ray scattering from solutions of nanoparticles demonstrates high sensitivity to orientational particle inhomogeneities.
BIOLOGY | MEDICINE
The tryptophan synthases from three human pathogens show remarkable structural conservation, but at the same time display local differences in both their catalytic and allosteric sites that may be responsible for the observed differences in catalysis and inhibitor binding. This functional dissimilarity may be exploited in the design of species-specific enzyme inhibitors.
BIOLOGY | MEDICINE
Both global and specific radiation damage have been investigated using three different proteins at cryogenic and room temperature. The large decoupling between global and specific radiation damage at cryogenic temperature appears to be practically abolished at room temperature, which has positive implications for time-resolved protein crystallography.
PDB references: Cerulean, cryogenic temperature structure, accumulated dose 290 kGy, 6qq8; cryogenic temperature structure, accumulated dose 5.8 MGy, 6qq9; room-temperature structure, accumulated dose 21 kGy, 6qqa; room-temperature structure, accumulated dose 147 kGy, 6qqb; hen egg-white lysozyme, cryogenic temperature structure, accumulated dose 110 kGy, 6qqc; cryogenic temperature structure, accumulated dose 10 MGy, 6qqd; room-temperature structure, accumulated dose 20 kGy, 6qqe; room-temperature structure, accumulated dose 100 kGy, 6qqf; AtPhot2LOV2, ground state, cryogenic temperature structure, accumulated dose 2.68 MGy, 6qqh; blue light-irradiated, cryogenic temperature structure, accumulated dose 24 kGy, 6qqi; ground state, room-temperature structure, accumulated dose 384 kGy, 6qqj; blue light-irradiated, room-temperature structure, accumulated dose 34 kGy, 6qqk; blue light-irradiated, cryogenic temperature structure, accumulated dose 48 kGy, 6qsa
CHEMISTRY | CRYSTENG
A method for the selective synthesis of a 2D layered MOF in the presence of the competitive formation of a 3D cubic MOF was developed. In addition, the laminated 2D MOF layers are directly synthesized via a modified bottom-up lamination method involving both chemical (surfactants) and physical (ultrasonication) stimuli.
MATERIALS | COMPUTATION
A large number of topological materials with a CsCl-type structure in crystallographic databases are explored and their crystal and electronic structures are studied.
CHEMISTRY | CRYSTENG
Exploration of a `time on shelf' protein structure containing the radiopharmaceutical synthon fac-[Re(CO)3(H2O)3]+ as an in vivo reaction vessel to form tetranuclear rhenium clusters appropriate for theranostic applications is reported.
CHEMISTRY | CRYSTENG
Download citation


Download citation


Radiation damage to small-molecule compounds during synchrotron-diffraction data collection, under a range of conditions, is reported, characterized and analysed.
CCDC reference: 1901775
BIOLOGY | MEDICINE
Direct protein crystallization on fixed-target sample holders allows highly efficient serial crystallography experiments and ligand-binding studies.
PDB references: proteinase K crystallized on a silicon chip, 6qf1; thermolysin crystallized on a silicon chip, 6qf2; thermolysin soaked with sodium aspartate on a silicon chip, 6qf3; human serine/threonine kinase 17B in complex with ADP obtained by on-chip soaking, 6qf4; human aquaporin 2 crystallized on a silicon chip, 6qf5
BIOLOGY | MEDICINE
Ependymin-related proteins fold into a dimer using a subunit formed by stacking antiparallel β-sheets and contain a hydrophobic pocket that is found in the similar folds of bacterial VioE and LolA/LolB. As in VioE and LolA/LolB, this hydrophobic pocket may be used to bind hydrophobic molecules.
CHEMISTRY | CRYSTENG
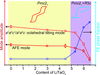
Symmetry-mode analysis has been used to construct the direct linkage between structure and properties for (anti)ferroelectric materials.
CHEMISTRY | CRYSTENG
Download citation


Download citation


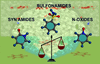
Crystal engineering of the sulfonamide group with competing coformer molecules (lactam, syn-amide and N-oxide) using MEP calculations suggests that the SO2NH2 group will bond to the lactam and syn-amide preferentially compared with N-oxide and carboxylic acid, but complexation studies showed superior bonding with N-oxide. These results provide a ranking of hydrogen-bonding synthons for crystal engineering with the sulfonamide group.
PHYSICS | FELS
Damage-free structures of the resting state of one of the most studied copper nitrite reductases were obtained independently by X-ray free-electron laser (XFEL) and neutron crystallography, representing the first direct comparison of neutron and XFEL structural data for any protein. In addition, damage-free structures of the reduced and nitrite-bound forms have been obtained to high resolution from cryogenically maintained crystals by XFEL crystallography.
PDB references: Achromobacter cycloclastes CuNiR, resting state, SF-ROX structure, 6gsq; resting state, neutron structure, 6gtj; pH 5, resting state, low dose, 6gti; pH 5.5, resting state, low dose, 6gtk; pH 6, resting state, low dose, 6gtl; pH 6.5, resting state, low dose, 6gtn; nitrite-bound, SF-ROX structure, 6gt0; chemically reduced, SF-ROX structure, 6gt2
CRYO | EM
The cryo-EM structure of Neurospora crassa respiratory complex IV was determined to 5.5 Å resolution and is compared with related structures from Saccharomyces cerevisiae and Bos taurus.
addenda and errata
BIOLOGY | MEDICINE
Corrigendum to the article by Dajnowicz et al. [IUCrJ (2019). 6, 619–629].



 journal menu
journal menu




 access
access