issue contents
January 2017 issue
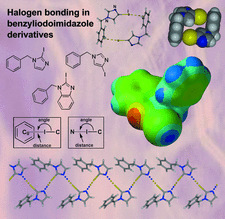
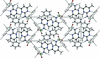
Cover illustration: The role of halogen bonding, i.e. C-I N and C-I
N and C-I
![[pi]](/logos/entities/pi_rmgif.gif) , as a major intermolecular force is investigated in the crystal structures of three iodoimidazole derivatives. See Nwachukwu, Bowling & Bosch [Acta Cryst. (2017), C73, 2-8].
, as a major intermolecular force is investigated in the crystal structures of three iodoimidazole derivatives. See Nwachukwu, Bowling & Bosch [Acta Cryst. (2017), C73, 2-8].
editorial
Free 

The evolution of Acta Crystallographica Section C is progressing steadily and features an increasing number of papers that include a broader spectrum of science in the discussion in addition to the crystal structure determinations. The Scopus CiteScore for the journal, which was 0.57 in 2015, is showing an interim value of 3.56 for 2016 at time of writing.
research papers
Download citation


Download citation


Free 

The role of halogen bonding, i.e. C—I⋯N and C—I⋯π, as a major intermolecular force is investigated in the crystal structures of three iodoimidazole derivatives.
Download citation


Download citation


The ability of sulfamethazine (SMZ) to form solvates has been investigated. Both pyridine and 3-methylpyridine form solvates with SMZ in 1:1 molar ratios. The solvent molecules in both structures play an active part in strong intermolecular interactions, thereby contributing significantly to the stability of both structures.
Download citation


Download citation


The crystals of two new salts in the DL-norvaline–maleic acid system provide examples of different molecular packing of the molecular building blocks, which are either the same, or differ merely by protonation.
Download citation


Download citation


In 1,2-O-cyclohexylidene-myo-inositol, the hydroxy groups at positions 4 and 5 are in a trans configuration and a rationale is suggested for the difficulties in synthesizing 1,2;4,5-di-O-cyclohexylidene-myo-inositol. The calculated structure was very similar to that of the experimental structure. Hirshfeld surface analysis with fingerprint plots demonstrated that the O—H⋯O interactions are the major intermolecular interactions. Lattice energy calculations suggested that dispersion is the major contributor to the crystal packing.
CCDC reference: 1518275
Download citation


Download citation


Three closely-related tetracyclic dibenzoazepine derivatives, two of them dibenzothiazoloazepinones and the third a benzoazepinoquinolone, all show different molecular conformations, different patterns of intermolecular hydrogen bonding and different modes of supramolecular aggregation. One crystallizes as a kryptoracemate in the space group P21, and the other two as true racemates in centrosymmetric space groups.
Download citation


Download citation


To explore the coordination behaviour of the allyl derivatives of thiadiazoles, such as 5-methyl-N-(prop-2-en-1-yl)-1,3,4-thiadiazol-2-amine (Mepeta) and 5-[(prop-2-en-1-yl)sulfanyl]-1,3,4-thiadiazol-2-amine (Pesta) with respect to diverse copper(I) salts, we report herein the syntheses and structures of five new π-complexes. The influence of the ligand coordination mode on the rare case of a CuI⋯FSiF52− bond is also highlighted.
Download citation


Download citation


The crystal structure of the coproporphyrin I RuII complex catena-poly[[carbonylruthenium(II)]-μ-2,7,12,17-tetrakis[2-(ethoxycarbonyl)ethyl]-3,8,13,18-tetramethylporphyrinato] has been determined from high-resolution synchrotron powder diffraction data. The RuII centre is coordinated by four N atoms in a basal plane, and by axial C and O atoms in a distorted octahedral geometry. Self-assembly of the molecules during crystallization gives one-dimensional polymeric chains.
CCDC reference: 1520660
Download citation


Download citation


The reactions of benzoyl isothiocyanate with 3,4-dimethoxyaniline and 1-aminopropan-2-ol yielded two novel 1-benzoylthiourea derivatives. In both structures, intramolecular O—H⋯N hydrogen bonding is present, forming six-membered pseudo-rings that are quasi-aromatic and interact with phenyl rings and with each other via stacking-type interactions. Intermolecular hydrogen bonds result in the formation of centrosymmetric dimers or one-dimensional chains. C—H⋯(O,S) interactions additionally reinforce the three-dimensional structure of both compounds.
Download citation


Download citation


Two new B,P,N-containing compounds were synthesized via simple reactions and were structurally characterized. Intermolecular hydrogen bonds link the independent molecules into supramolecular networks.
Download citation


Download citation


In the series of hydrogen-bonded structures of the alkaline earth metal (Mg, Ca, Sr and Ba) complexes with (4-aminophenyl)arsonic acid, with the exception of the Mg salt, all members form one-dimensional coordination polymers. Hydrogen-bonding interactions give overall three-dimensional supramolecular structures which, in the case of the Sr and Ba complexes, include π–π ring interactions.


 journal menu
journal menu