issue contents
April 2020 issue
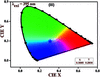
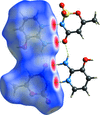
Cover illustration: 4-[(Morpholin-4-yl)carbothioyl]benzoic acid, C12H13NO3S, a novel phenyl (morpholino)methanethione derivative, represents the first crystal structure for which all experimental work (synthesis, crystallization, data collection, structure solution and refinement) was performed in Benin. This important result for Crystallography in Africa is a product of the first crystallographic training session at the X-TechLab in Sèmè City, organized within the IUCr-UNESCO OpenLab framework. See: Agnimonhan, Bendeif, Akanni, Gbaguidi, Martin, Wenger & Lecomte [Acta Cryst. (2020). E76, 324-327].
research communications
Download citation


Download citation


The title compound consists of pyrrole and benzodiazepine units linked to a propargyl moiety, where the pyrrole and diazepine rings adopt half-chair and boat conformations, respectively. In the crystal, weak C—HBnz⋯ODiazp and C—HProprg⋯ODiazp (Bnz = benzene, Diazp = diazepine and Proprg = propargyl) hydrogen bonds link the molecules into two-dimensional networks parallel to the bc plane, enclosing 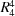 (28) ring motifs.
(28) ring motifs.
CCDC reference: 1986475
Download citation


Download citation


In the crystal of the title compound, the packing is driven by O—H⋯O, O—H⋯N and N—H⋯N hydrogen-bond interactions along with π–π stacking interactions.
CCDC reference: 1986751
Download citation


Download citation


The crystallization of (E)-1-(5-chlorothiophen-2-yl)-3-(3-methylthiophen-2-yl)prop-2-en-1-one furnished a superimposed co-crystal consisting of the expected chalcone and a minor component identified as (Z)-1-(5-chlorothiophen-2-yl)-3-(3-methylthiophen-2-yl)prop-1-en-1-ol tautomer
CCDC reference: 1986074
Download citation


Download citation


In the title compound, C13H15N3O5S, the two rings face each other in a `V′ form at the S atom, with one N—H⋯O=S and one C—H⋯O=S contact from the pyrazolyl substituents to the sulfonyl group. Two classical hydrogen bonds from the amine group, one of the form N—H⋯O=S and one N—H⋯O=Coxo, link the molecules to form layers parallel to the bc plane.
CCDC reference: 1986369
Download citation


Download citation


The title compound is an example of an ortho-vanillin-based functionalized dithioether, which could be useful as a potential chelating ligand or bridging ligand for coordination chemistry. Its crystal structure was determined at 100 K. Both weak intramolecular O—H⋯O and intermolecular O—H⋯S hydrogen bonding can be observed.
CCDC reference: 1983985
Download citation


Download citation


The hydrogen-bonded assemblies in N-(4-methoxyphenyl)piperazine and its 3,5-dinitrobenzoate, picrate, and monohydrated 4-aminobenzoate salts are, respectively, one-, zero-, two- and three-dimensional.
Download citation


Download citation


In the title hydrated complex, [Co(C15H10ClN3)2]Cl2·4H2O, the complete dication is generated by  symmetry. In the crystal, O—H⋯Cl and C—H⋯O hydrogen bonds link the components into (001) sheets. The complex exhibits blue-light emission.
symmetry. In the crystal, O—H⋯Cl and C—H⋯O hydrogen bonds link the components into (001) sheets. The complex exhibits blue-light emission.
CCDC reference: 1914486
Download citation


Download citation


A one-dimensional nickel(II) coordination polymer with the mixed ligands 6-fluoronicotinate (6-Fnic) and 4,4′-bipyridine (4,4′-bpy), namely, [Ni(H2O)2(6-Fnic)2(4,4′-bpy)·3H2O]n, (1), was prepared. The nickel(II) ion in 1 is octahedrally coordinated by the O atoms of two water molecules, two O atoms from O-monodentate 6-fluoronicotinate ligands and two N atoms from bridging 4,4′-bipyridine ligands.
CCDC reference: 1988000
Download citation


Download citation


In the title molecular salt, (C7H9N4O2)[ZnCl3(H2O)], the crystal packing exhibits O—H⋯O, O—H⋯Cl, N—H⋯O and N—H⋯Cl hydrogen bonds.
CCDC reference: 1986654
Download citation


Download citation


The crystal structures of praseodymium silicide (5/4), Pr5Si4, and neodymium silicide (5/4), Nd5Si4, were redetermined at higher precision. These compounds are confirmed to have the tetragonal Zr5Si4-type structure (space group: P41212). The structure is built up by distorted body-centered cubes consisting of Pr(Nd) atoms, which are linked to each other by edge-sharing to form a three-dimensional framework. Silicon dimers are located within channels in this framework.
Download citation


Download citation


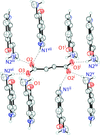
A new form of the synthetic psychedelic miprocin, a psilocin derivative, is reported. The title compound has a single protonated tryptammonium cation and one half of a fumarate in the asymmetric unit. The ions are held together in an elaborate set of rings and chains along (110).
CCDC reference: 1987588
Download citation


Download citation


The redetermined structure of the title compound, [H4N][C6H11Mo2O11]· H2O, was obtained from an attempt to prepare a glutamic acid complex from the [Co2Mo10H4O38]6− anion.
CCDC reference: 1965028
Download citation


Download citation


The asymmetric unit of the title compound, (C14H36N4)[CrO3Cl]2Cl2, contains one half-cation (completed by crystallographic inversion symmetry), one chlorochromate anion and one chloride anion. In the crystal, N—H⋯Cl, C—H⋯Cl and C—H⋯O hydrogen bonds connect the organic cations, chlorochromate and chloride anions, forming a three-dimensional network.
CCDC reference: 1988052
Download citation


Download citation


The title molecular salts containing the same cation accompanied by different simple anions show quite different intermolecular connectivities.
Download citation


Download citation


The title compound forms triclinic crystals in which the methylpiperidine ring is in its stable chair conformation. The dihedral angle between the mean planes of the benzene ring and that of the twisted piperidine ring is 39.89 (7)°. In the crystal, weak C—H⋯O interactions link the molecules into infinite chains along the a-axis direction.
CCDC reference: 1973841
Download citation


Download citation


On reaction with silver nitrate the new pyrazinethiophanes, 5,7-dihydro-1H,3H-dithieno[3,4-b:3′,4′-e]pyrazine and 3,4,8,10,11,13-hexahydro-1H,6H-bis([1,4]dithiocino)[6,7-b:6′,7′-e]pyrazine, both form two-dimensional coordination polymers.
Download citation


Download citation


Solid-state structures of the title compounds are presented to show the relative effects of 1,3,5-triaza-7-phosphaadamantane (PTA) and 3,7-diacetyl-1,3,7-triaza-5-phosphabicyclo[3.3.1]nonane (DAPTA) ligands on the molecular and extended structure.
Download citation


Download citation


In both structures, the alkyloxy and methylsulfanyl groups are rotated out of the plane of the respective heterocyclic ring.
Download citation


Download citation


The hydrazide-hydrazone forms inverse dimers via hydrogen bonding, but its conformation is defined by the presence of an intramolecular chalcogen bond. Electrostatic forces dominate in the crystal packing and give rise to a layered supramolecular structure.
CCDC reference: 1983191
Download citation


Download citation


Methionine hydroxy analogue, a common poultry feed supplement, has been obtained in crystalline form for the first time. The asymmetric unit contains two conformationally unequal molecules that are involved in a two-dimensional intermolecular hydrogen-bonding network.
CCDC reference: 1979735
Download citation


Download citation


The structural analysis of a phenylindolizine-based drug, namely methyl 3-(4-flurobenzoyl)-7-methyl-2-phenylindolizine-1-carboxylate (I) was carried out; this drug shows an inhibitory action towards mycobacterium tuberculosis. The intermolecular interactions at play were characterized via Hirshfeld surface analysis and fingerprint plots, highlighting the evident role of C—H⋯O, C—H⋯F and C—H⋯π interactions in the formation of the observed crystal structure.
CCDC reference: 1865697
Download citation


Download citation


In the crystal of the title compound, C9H11N3O5, the 2-amino-6-hydroxypyridin-1-ium cations and 6-methyl-2,2,4-trioxo-2H,4H-1,2,3-oxathiazin-3-ide anions are held together through N—H⋯O, N—H⋯N, O—H⋯O and C—H⋯O hydrogen bonds.
CCDC reference: 1920140
Download citation


Download citation


The dihedral angle between the mean plane of the anthraquinone ring system and the dioxepine ring in the title compound is 16.29 (8)°. The packing is consolidated by C—H⋯O, π–π and C=O⋯π interactions.
CCDC reference: 1991271
Download citation


Download citation


The first molecular compound synthesized and crystallized in Benin is reported. This work was carried out during the first crystallographic training session at the X-TechLab in Sèmè City, organized within the IUCr–UNESCO OpenLab framework.
CCDC reference: 1991487
Download citation


Download citation


In the acridinedione moiety of the title compound, the central ring adopts a flattened-boat conformation, whereas the cyclohexenone rings adopt envelope conformations.
CCDC reference: 924670
Download citation


Download citation


The solid-state structure of the `magic mushroom' natural product norpsilocin is reported, as well as the synthesis and structure of its fumarate salt.
Download citation


Download citation


In the title compound, the amide functional group –C(=O)NH– adopts a trans conformation with the four atoms nearly coplanar. This conformation promotes the formation of a C(4) hydrogen-bonded chain propagating along the [010] direction.
CCDC reference: 1987798


 journal menu
journal menu