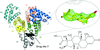issue contents
November 2020 issue
scientific commentaries
PHYSICS | FELS
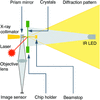
A new program, diffBragg, employs per-pixel maximum likelihood optimization of X-ray pulse and crystal parameters to improve the accuracy of structure factor amplitudes attainable in SFX experiments.
MATERIALS | COMPUTATION
After a hundred years with no solution, the seemingly simple but actually very complex mineral kaliophilite, KAlSiO4, is finally revealed by electron crystallography.
BIOLOGY | MEDICINE
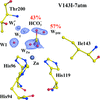
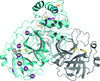
Kim and co-workers [IUCrJ (2020). 7, 985–994] advance our understanding of the catalytic mechanism of carbonic anhydrase II by studying a mutant V143I where the change (of one hydrophobic amino acid to another that differs by a single CH2 group) is probably the smallest alteration that can be introduced into a protein. The study was performed at high pressure in a CO2 atmosphere to visualize the bound substrate; it showed the behavior of the entrance conduit waters and the substrate alteration due to the mutation.
topical reviews
CHEMISTRY | CRYSTENG
The rich solid-state chemistry of olanzapine uncovered during the development of this blockbuster drug has inspired the development and application of new tools and techniques for understanding molecular assembly during crystallization.
research letters
PHYSICS | FELS
Significant improvements to the current state-of-the-art in phasing strategies in serial femtosecond crystallography are presented and quantified.
PDB references: A2A, 4.57 keV, all diffraction images, 6s0l; A2A, 4.57 keV, 50 000 diffraction images, 6s0q; thaumatin, 4.57 keV, all diffraction images, 6s19; thaumatin, 4.57 keV, 20 000 diffraction images, 6s1d; thaumatin, 6.06 keV, all diffraction images, 6s1e; thaumatin, 6.06 keV, 50 000 diffraction images, 6s1g
PHYSICS | FELS
Serial femtosecond crystallography of adenosine A2A receptor (A2AAR) crystallized in lipidic cubic phase was performed using high X-ray free-electron laser transmission with a high dynamic range detector in a helium atmosphere. The 2.0 Å resolution A2AAR structure model is presented and compared with previous A2AAR structures determined in a vacuum and/or at cryogenic temperatures.
research papers
BIOLOGY | MEDICINE
X-ray crystallography was used to elucidate the effect of a single-site mutation on the activity of a native metalloenzyme. The subtle structural modifications around the active site of the enzyme were correlated with the retarded catalytic efficiency in terms of the mechanistic steps and their kinetics.
CRYO | EM
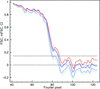
A modified Fourier shell correlation methodology is described that permits a robust, objective and reproducible assessment of resolution in single-particle cryo-EM, including helical assemblies. The method eliminates the adverse influence of masking the results. The inclusion of statistical error analysis mitigates common problems with resolution overestimation. It is shown that the method is also applicable to local resolution estimation and to resolution assessment of secondary elements such as helices and β-sheets.
BIOLOGY | MEDICINE
Download citation


Download citation


An automated platform for diffraction experiments at room temperature for the study of macromolecular structures and dynamics with important biological significance is reported here. This platform preserves the virgin quality of crystal samples and requires low sample consumption.
BIOLOGY | MEDICINE
Structures of monothiol and dithiol glutaredoxin-1 from C. oremlandii are reported. Monothiol glutaredoxin-1 formed domain-swapped structures, one of which was in a hetero-oligomer complex.
BIOLOGY | MEDICINE
The viral main protease is indispensable for SARS-CoV-2 replication, and detailed knowledge of its structure and function is crucial to guide structure-aided and computational drug-design efforts. X-ray crystallography is used to reveal the oxidation and reactivity of the cysteine residues of the protease, providing insights into the enzyme mechanism and drug design.
PDB references: SARS-CoV-2 3CL Mpro, peroxysulfenic Cys145 structure (structure I), 6xb0; peroxysulfenic Cys145/N-ethylmaleimide Michael addition Cys156 structure (structure II), 6xb1; Cys145/Cys156 N-ethylmaleimide Michael addition structure (structure III), 6xb2; native structure at pH 6.0 (structure IV), 6xhu
MATERIALS | COMPUTATION
Twenty new Nd–Fe–B crystal structures can be found via the elemental substitution of 5967 host structures of lanthanides–transition metals–light elements collected from the Open Quantum Materials Database. The average atomic coordination number and coordination number of the Fe sites are the most important factors in determining the structure–stability relationship of the new substituted Nd–Fe–B crystal structures.
BIOLOGY | MEDICINE
The structure of serum albumin in complex with dexamethasone reveals why the drug may not always help COVID-19 patients.
PDB reference: serum albumin, complex with dexamethasone, 6xk0
CRYO | EM
Download citation


Download citation


The flexibility and conformational dynamics of the SARS-CoV-2 spike in the prefusion state have been analyzed. An ensemble map with minimum bias was obtained, revealing concerted motions involving the receptor-binding domain (RBD), N-terminal domain and subdomains 1 and 2 around the previously characterized 1-RBD-up state, which have been modeled as elastic deformations.
EMDB references: SARS-CoV-2 spike in prefusion state, EMD-11328; SARS-CoV-2 spike in prefusion state (flexibility analysis, 1-up closed conformation), EMD-11336; SARS-CoV-2 spike in prefusion state (flexibility analysis, 1-up open conformation), EMD-11337; SARS-CoV-2 stabilized spike in prefusion state (1-up conformation), EMD-11341
MATERIALS | COMPUTATION
Download citation


Download citation


The elusive structure of the mineral kaliophilite has been determined by 3D electron diffraction and refined using single-crystal X-ray data. Despite its simple formula, ideally KAlSiO4, the structure of this mineral remained a mystery for over a century as a result of pseudo-symmetry and twinning, which reduce the coherent crystalline domain size to a few hundreds of nanometres.
CCDC reference: 2027405
BIOLOGY | MEDICINE
Novel approaches for lipid sponge phase crystallization in comparatively large volumes using Hamilton gas-tight glass syringes and plastic pipetting tips are described. The selection of distinct co-crystallization conditions allowed to restore one of the natural ligands displaced by monoolein to its binding site.
BIOLOGY | MEDICINE
Crystal structure determination of membrane proteins can be challenging. Here, the use of vanadium phasing as an alternative experimental phasing method is described.
PHYSICS | FELS
The 3D structure of bacteriophage PR772 was determined from a single-particle imaging experiment at the Linac Coherent Light Source, using the limited number of data, with a spatial resolution of 6.9 nm.
PHYSICS | FELS
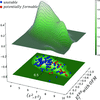
Single-particle imaging using X-ray free-electron lasers requires an intensive X-ray fluence which damages the sample as it is imaged. This paper explores the potential for including theoretical simulations of single-particle electrodynamics into a reconstruction of the electron density imaged using X-ray free-electron lasers. While partial coherence, induced by radiation damage, poses a challenge for phase retrieval, it is demonstrated that additional coherent modes enable suppression of the effect of radiation damage in the reconstructed sample electron density.
CHEMISTRY | CRYSTENG
Download citation


Download citation


In order to form a solid solution with its prodrug cortisone, hydrocortisone (cortisol) must violate Etter's rule of hydrogen bonding. The preparation of a uniform product with the desired composition, which is hindered under a thermodynamic regime, can be achieved by supercritical assisted spray drying. The new phase enables fine-tuning of the phase's composition as well as a higher dissolution rate for hydrocortisone.
NEUTRON | SYNCHROTRON
A microcrystal-prelocation method is demonstrated using low-dose 2D full-field propagation-based X-ray phase-contrast imaging on samples with an essentially flat geometry for automated serial crystallography data collection at a microfocus macromolecular crystallography beamline.
PDB references: lysozyme from SiN IMISX setup collected by rotation serial crystallography, 6yod; lysozyme from SiN IMISX setup collected by still serial crystallography, 6yoe; PepTSt from COC IMISX setup collected by rotation serial crystallography, 6yof; lysozyme from COC IMISX setup collected by rotation serial crystallography, 6yob; lysozyme from COC IMISX setup collected by still serial crystallography, 6yoc; PepTSt from COC IMISX setup collected by still serial crystallography, 6yog
CRYO | EM
It is demonstrated that a convolutional neural network denoising algorithm can be used to significantly enhance the signal-to-noise ratio and generate contrast in cryo-EM images. It also provides a quantitative evaluation of the bias introduced by the denoising procedure and its influence on image processing and three-dimensional reconstructions.
PHYSICS | FELS
A pixel-based approach for extracting structure factors from X-ray free-electron laser crystal diffraction measurements is presented.
BIOLOGY | MEDICINE
This paper describes and evaluates the performance of AWSEM-Suite, an algorithm that includes template-guided refinement and coevolutionary information within the framework of the energy-landscape theory, in using molecular replacement to solve the phase problem by the de novo prediction of structures. It also highlights and discusses how AWSEM-Suite provides better predictions than I-TASSER-MR or the previous algorithm AWSEM-Template.
CRYO | EM
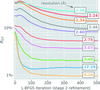
In this work, it is shown that significant microscope aberrations caused by beam-image-shift cryo-EM at 200 keV can be overcome using computational correction, improving a structure of aldolase from 4.9 to 2.8 Å resolution.
CHEMISTRY | CRYSTENG
Download citation


Download citation


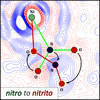
A new, cheap, easy-to-synthesize and air-stable photoswitchable nickel nitro complex, in which the central metal atom is additionally chelated by a [2-methyl-8-aminoquinoline]-1-tetralone ligand, is reported. The compound was shown to undergo full light-induced isomerization from the nitro to nitrito form by irradiation with 530–660 nm LED light at 160 K, whereas the generated linkage isomer exhibits thermal stability up to around 230 K.
CHEMISTRY | CRYSTENG
In this work, various models of atomic electron density were applied in a generalized version of the Hirshfeld atom refinement to three organic structures.
CCDC references: 2040066; 2042674; 2042675; 2042676; 2042677; 2042678; 2042679; 2042680; 2042681; 2042682; 2042683; 2042684; 2042685; 2042686; 2042687; 2042688; 2042689; 2042690; 2042691; 2042692; 2042693; 2042694; 2042695; 2042696; 2042697; 2042698; 2042699; 2042700; 2042701; 2042702; 2042703; 2042704; 2042705; 2042706; 2042707; 2042708; 2042709; 2042710; 2042711; 2042712; 2042713; 2042714



 journal menu
journal menu




 access
access