issue contents
April 2019 issue
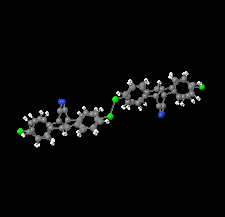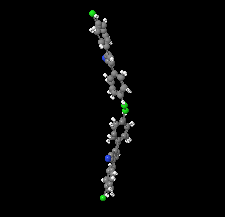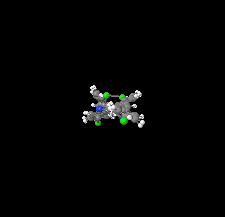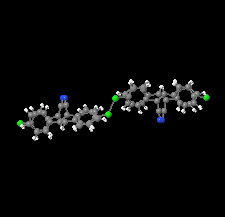
Cover illustration: A new acrylonitrile derivative, namely (Z)-3-(4-chlorophenyl)-2-phenylacrylonitrile, has been synthesized and characterized in the solid state in the framework of a research project that focuses on the optical properties of this class of materials, as well as on their versatility as building blocks in supramolecular assemblies. The crystal structure of the title compound is mainly directed by weak halogen and hydrogen bonds, along with C—H⋯π interactions; the resulting packing has been studied in detail through Hirshfeld surface analysis, while lattice and intermolecular interaction energies have been determined using the PIXEL method. This has allowed the contributions of electrostatic forces, polarization, dispersion and exchange-repulsion to the total energy of the system to be identified and quantified, and their role in the stabilization of the overall crystal structure to be investigated. See: Udayakumar, Cerón, Ceballos, Percino & Thamotharan [Acta Cryst. (2019). E75, 499–505].
research communications












































 journal menu
journal menu