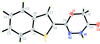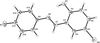issue contents
January 2014 issue
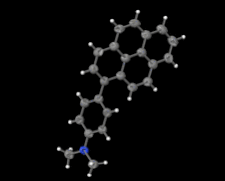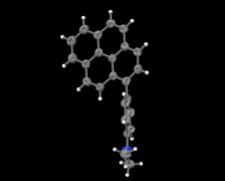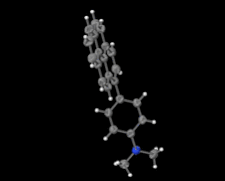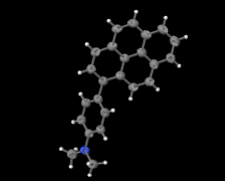
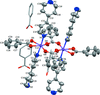
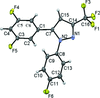
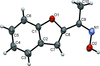
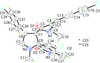
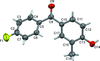
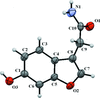
Cover illustration: Electron donor–acceptor molecules play an important role in the understanding of charge-transfer processes. In the compound C24H19N, the electron donor N,N-dimethylaniline (DMA) is covalently linked to the electron acceptor pyrene (Py). The structural and physical characteristics of these donor and acceptor groups strongly influence the electron-transfer mechanism, which leads to unusual absorption and emission spectra in the optical regime of PyDMA, making it a molecular diode in which electron-donor and electron-acceptor moieties are twisted against each other, thus modulating the electron charge-transfer processes. The dimethylamino group is inclined to the benzene ring by 2.81 (9)°. The mean plane through these groups makes a dihedral angle of 64.12 (2)° with the mean plane of the pyrene ring system. See: Thekku Veedu, Scholz, Kia, Paulmann & Techert [Acta Cryst. (2014). E70, o16].
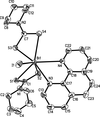
inorganic compounds










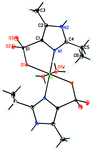
metal-organic compounds








































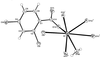
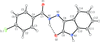
organic compounds

















































































































































 journal menu
journal menu