issue contents
March 2020 issue
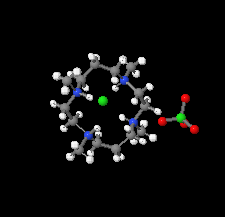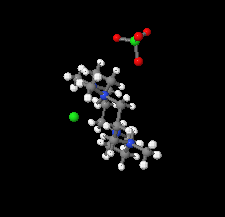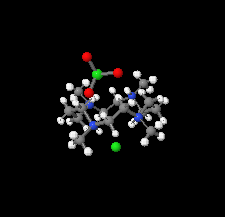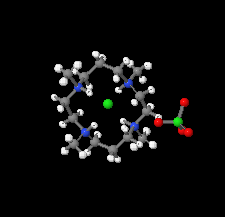
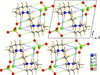
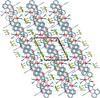
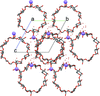
Cover illustration: Tetraazamacrocycles are useful in a variety of applications, in particular in the elimination of toxic metals. The N-substituted tetraazamacrocyle discussed in this contribution (1,4,8,11-tetramethyl-1,4,8,11-tetraazoniacyclotetradecane) is a tetracation resulting from the reaction of 1,4,8,11-tetramethyl-1,4,8,11-tetraazacyclotetradecane (TMC, C14H32N4) with sodium perchlorate/chromium trioxide in HCl. The structure determination by synchrotron radiation indicated that this tetra-protonated amine displays a distorted trans-IV conformation, one of the five possible conformations of the macrocyclic cation. The structure is characterized by an intricate and extensive network of N-H Cl and C-H
Cl and C-H Cl hydrogen bonds, which link the two Cl- anions to the C14H36N44+ cation, while C-H
Cl hydrogen bonds, which link the two Cl- anions to the C14H36N44+ cation, while C-H O hydrogen bonds interconnect neighboring cations with the perchlorate anions. See: Moon & Choi [Acta Cryst. (2020). E71, 324-327].
O hydrogen bonds interconnect neighboring cations with the perchlorate anions. See: Moon & Choi [Acta Cryst. (2020). E71, 324-327].
research communications




































































 journal menu
journal menu