issue contents
June 2015 issue
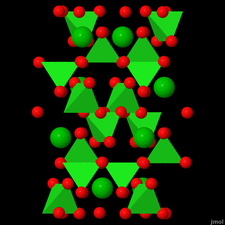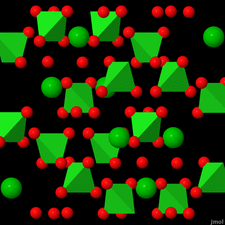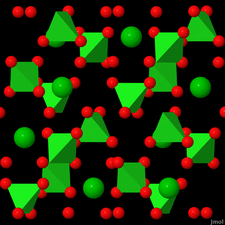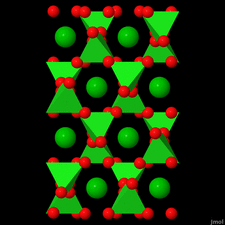
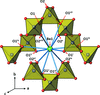
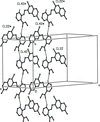
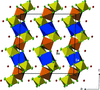
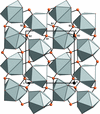
Cover illustration: Anhydrous alkaline-earth perchlorates have important uses in electrochemistry, but due to the difficulty in growing single crystals, little is known about their structures. Lee, Kang, Lim & Hong [Acta Cryst. (2015). E71, 588-591] dehydrated a sample of [Ba(ClO4)2·xH2O] and determined the structure of Ba(ClO4)2 ab initio from laboratory powder X-ray data to reveal a new structure type built up from corner- and edge-sharing BaO12 and ClO4 polyhedra.
research communications
Download citation


Download citation


The title compound has an E conformation with respect to the azomethine C=N bond and the aromatic rings are inclined to one another by 3.29 (4)°. In the crystal, molecules are linked via C—H⋯O hydrogen bonds, forming zigzag chains along [10-1].
CCDC reference: 1062128
Download citation


Download citation


Three thiochromeno[4,3-b]pyrrole esters have very similar conformations. Structurally two of the compounds differ only by the substituent on the benzene ring, i.e. 4-isopropylphenyl and o-tolyl, while two of the compounds differ only in that one has a pyrrole ring and one has an isoxazole ring.
Download citation


Download citation


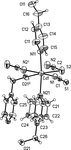
In the title compound, the Ni2+ cation is coordinated by two deprotonated amido N atoms and two carboxylate O atoms, setting up a square-planar coordination environment. The cations and the anion are linked by weak intra- and intermolecular C—H⋯O and C—H⋯F hydrogen bonds.
CCDC reference: 1062184
Download citation


Download citation


The structures of three disaccharide molecules, precursors to novel therapeutics, as determined from weakly diffracting crystals are presented. The crystal packing depends mainly on weak C—H⋯O hydrogen-bond interactions, augmented by C—H⋯π contacts in the best-defined structure.
Download citation


Download citation


The previously unknown crystal structure of barium perchlorate anhydrate, determined and refined from laboratory X-ray powder diffraction data, represents a new structure type. The structure can be described as a three-dimensional polyhedral network resulting from the corner- and edge-sharing of BaO12 polyhedra and ClO4 tetrahedra.
CCDC reference: 1063587
Download citation


Download citation


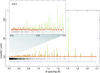
High-precision structural parameters for cubic Na2MoO4 and Na2WO4 are reported based on refinement of high-resolution time-of-flight neutron powder diffraction data. Complementary Raman spectra are also provided.
Download citation


Download citation


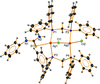
The anionic framework of (Na0.4,Li0.6)(Fe,Li2)(MoO4)3 is built up from two distinct MO6 octahedra, each containing disordered Li+ and Fe3+ ions, and two MoO4 tetrahedra, which link by vertex-sharing of their O atoms. These tetrameric units are further linked by sharing edges between octahedra and by formation of M—O—Mo (M = Fe/Li) bridges, forming ribbons propagating in the [100]. The ribbons are cross-linked in both the b- and c-axis directions, giving rise to a three-dimensional framework having [100] tunnels in which the monovalent Na+/Li+ cations lie.
CCDC reference: 1063336
Download citation


Download citation


The title arylsulfonyl glycinyl hydrazone Schiff base compound crystallizes as a monohydrate. In the crystal, a series of O—H⋯O and N—H⋯O hydrogen bonds leads to the formation of corrugated sheets lying parallel to (100).
CCDC reference: 1062518
Download citation


Download citation


The crystal stucture of 3-amino-4-nitrobenzyl displays intramolecular resonance-assisted hydrogen bonding between the ortho amino and nitro groups in addition to an intermolecular network of hydrogen bonding and π-stacking.
CCDC reference: 1063364
Download citation


Download citation


Five closely related (2-chloroquinlin-3-yl)methyl ethers all exhibit different patterns of direction-specific intermolecular interactions, leading to the formation of different types of chain in four of them and sheets in the fifth.
Download citation


Download citation


The title molecular salt, consists of a sulfamethoxazolium (SMZ) cation and a 3,5-dinitrosalicylate (DNS) anion, which are linked by an N—H⋯O hydrogen bond. In the crystal, the cations and anions are linked via N—H⋯O, N—H⋯N and C—H⋯O hydrogen bonds, forming a three-dimensional framework.
CCDC reference: 1063245
Download citation


Download citation


In the cation of the title molecular salt, the N atom is protonated with sp3-hybridization and has a tetrahedral geometry. In the crystal, the cations are bridged by the Cl− anions via N—H⋯Cl hydrogen bonds, forming four-centred inversion dimers with an 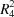 (8) ring motif.
(8) ring motif.
CCDC reference: 299500
Download citation


Download citation


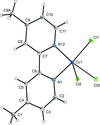
The structure of a previously unknown form of dichlorido(4,4′-dimethyl-2,2′-bipyridine)copper(II) was obtained via a DMSO-mediated dehydration of Cu(4,4′-dimethyl-2,2′-bipyridine)copper(II)·0.25H2O. The crystal structure reveals chloride-bridged copper(II) chains connected via intermolecular C—H⋯Cl hydrogen bonds.
CCDC reference: 1063931
Download citation


Download citation


To investigate the predominant intermolecular interactions in 1-alkyl-4-aryl-1,2,4-triazol-1-ium halide salts, five salts were prepared and crystallographically characterized. The halide ions generally interact with the H atoms of the triazolium cation forming extended sheets. When the aryl ring lies on the plane of the triazolium cation, the cationic core formed two-dimensional networks that lead into layer-like assembled structures. The triazolium core exhibits π–π interactions with the iodide and/or the aryl ring of another layer. The melting-point temperatures of each salt were also determined.
Download citation


Download citation


The structure of CsCrAs2O7 can be described as a three-dimensional [CrAs2O7]− anionic framework in which the Cs+ cations are located in empty channels running along [001].
CCDC reference: 1400446
Download citation


Download citation


The compound with the idealized composition Ba2CaFeAl2F14 crystallizes in the usovite structure type. Two models with different treatment of the disordered Fe site are presented.
Download citation


Download citation


The packing of the title compound features N—H⋯Cl hydrogen bonds and π–π stacking interactions, which form one-dimensional chains of molecules parallel to [001] further linked via N—H⋯O interactions.
CCDC reference: 1400786
Download citation


Download citation


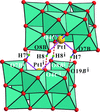
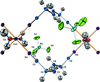
The polyanion in K5[H2PtV9O28]·9H2O has approximate mm2 (C2v) symmetry. The two platinum-bound μ2-O atoms are protonated in the polyanion. The heteropolyanions form inversion-generated dimers, {[H2PtV9O28]2}10−, held together by μ2-O—H⋯μ2-O and μ2-O—H⋯μ3-O hydrogen bonds.
CCDC reference: 1400654
Download citation


Download citation


The centrosymmetric CrIII ion in the title compound shows a distorted octahedral coordination with four N atoms of two ethane-1,2-diamine ligands in the equatorial plane and two N-coordinated NCS− groups in trans-axial positions. The ethane-1,2-diamine ligand in the complex cation and the ClO4− anion are both disordered.
CCDC reference: 1400767
Download citation


Download citation


The crystal structures of four indole derivatives with various substituents at the 2-, 3- and 5-positions of the ring system are described. The dominant intermolecular interaction in each case is an N—H⋯O hydrogen bond, which generates either chains or inversion dimers. Weak C—H⋯O, C—H⋯π and π–π interactions occur in these structures but there is no consistent pattern amongst them. Two of these compounds act as modest enhancers of CB1 cannabanoid signalling and two are inactive.
Download citation


Download citation


In the crystal structure of 1-[(6-chloropyridin-3-yl)sulfonyl]-1,2,3,4-tetrahydroquinoline, the tetrahydropyridine ring of the quinoline system adopts a half-chair conformation and the bond-angle sum at the N atom is 350.0°.
CCDC reference: 1061311
Download citation


Download citation


The structure of (NH4)[La(SO4)2(H2O)] comprises LaO9 polyhedra and SO4 tetrahedra, which are linked by common edges and vertices, forming a three-dimensional network with the hydrogen-bonded NH4+ ions in the cavities.
CCDC reference: 1401662
Download citation


Download citation


The N-[3-(dimethylamino)propyl]-N′-(2-hydroxyphenyl)oxamide trianion bridges two CuII cations to form the binuclear complex, in which the CuII cations have distorted square-planar and square-pyramidal coordination geometries.
CCDC reference: 1401557
Download citation


Download citation


The anhydrous tryptaminium salts of isomeric (2,4-dichlorophenoxy)acetic acid and (3,5-dichlorophenoxy)acetic acid give one-dimensional hydrogen-bonded chain structures which differ both in their cation–anion conformations and modes of inter-ion interaction.
Download citation


Download citation


Reactions of L- and DL-proline with CaI2 yields two crystalline products. The zwitterionic proline bridges the Ca cations with its carboxylate group in different coordination modes to form one-dimensional coordination polymers.
Download citation


Download citation


The crystal structure of 1,1,2,2-tetrachloroethane (TCE)-solvated 1,13,14-triazadibenz[a,j]anthracene (dibenzo[c,h]-1.9,10-anthyridine, dbanth) was determined by X-ray diffraction study. Two H atoms in the solvated TCE molecule form intermolecular C—H⋯(N,N) hydrogen bonds with three N atoms in dbanth. π–π interactions link the dbanth molecules to form a one-dimensional columnar structure.
Download citation


Download citation


In the title salt, [Zn(C8H4NO2)(C6H6N2O)(H2O)3](C8H4NO2), intermolecular O—H⋯O hydrogen bonds link two of the coordinating water molecules to two free 4-cyanobenzoate anions. N—H⋯O, O—H⋯O and C—H⋯O hydrogen bonds link the molecular components, enclosing 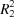 (12),
(12), 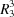 (8) and
(8) and 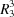 (9) ring motifs and forming layers parallel to (001).
(9) ring motifs and forming layers parallel to (001).
CCDC reference: 1401948
Download citation


Download citation


During synthesis of the corresponding aldehyde, 4-(trimethylgermyl)benzoic acid was obtained as a side-product. It crystallizes with two independent molecules in the asymmetric unit which exhibit slightly different geometries. In the crystal structure, centrosymmetric hydrogen-bonded dimers of the molecular pairs are formed.
CCDC reference: 1400647
Download citation


Download citation


The transition-metal orthophosphate Na1.67Zn1.67Fe1.33(PO4)3 cristallizes in an alluaudite-type structure. The chains characterizing the alluaudite structure are then built up from [M2O10] (M = Fe/Zn) units alternating with [ZnO6] octahedra. This structure is characterized by a cationic disorder in one tunnel and in the general position.
CCDC reference: 1402019
Download citation


Download citation


Two new nickel and copper tricyanomethanide (tcm−) complexes with 1,4,7,10-tetraazacyclododecane (cyclen) as a co-ligand have been synthesized and structurally characterized.
Download citation


Download citation


The title compound, [Ag2(CN)3(C9H8N2)2], is a mixed-valence disilver molecular complex. The Ag+ ion has the expected linear coordination geometry, while the Ag2+ centre is six-coordinated with a distorted [AgN5C] octahedral geometry. This compound belongs to class 1 or class 2 complexes in the Robin–Day classification.
CCDC reference: 1401857
Download citation


Download citation


The title compound, [MoO2(C14H9N2O2Br)H2O]·C3H7NO, has a three-dimensional supramolecular arrangement via a number of intermolecular C—H⋯O, O—H⋯N and O—H⋯O hydrogen bonds, as well as C—H⋯π and π–π interactions.
CCDC reference: 1401828
Download citation


Download citation


In the title complex, the CuII atom exhibits a square-planar coordination geometry and is located on a crystallographic inversion centre, leading to a trans configuration of the N,S-chelating ligands.
CCDC reference: 1057813
Download citation


Download citation


The binuclear coordination polymer consists of two nickel cations with different coordination environments. One has a square-planar environment whereas the other has an octahedral environment. Cyanide ligands bridge the cations into a polymeric layer structure.
CCDC reference: 1058383
Download citation


Download citation


Molecules of 1-ethyl-5-iodoindolin-2-one are arranged in columns extending along the a axis and interact with the molecules in adjacent columns via intermolecular C—H⋯O hydrogen bonds and I⋯I short contacts. A one-dimensional zigzag iodine chain along the a axis can be recognized between two neighbouring columns.
CCDC reference: 1028641
Download citation


Download citation


In the title salt, [Cu(C4H12P2S2)2]BF4, both diphosphine disulfide molecules bind to the CuI atom as chelating ligands via the S atoms, forming a monovalent cation with a slightly distorted tetrahedral coordination around the CuI atom. The ligand chelate rings are twisted in opposite directions, with one in the λ and one in the δ configuration. In the crystal, possible C—H⋯F hydrogen bonds may stabilize the orientation.
CCDC reference: 1402327
Download citation


Download citation


In the title indole derivative, the chlorophenyl ring is almost perpendicular to the indole moiety, making a dihedral angle of 87.59 (6)°. In the crystal, molecules are linked via C—H⋯π interactions, forming C(9) chains along the [10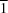 ] direction.
] direction.
CCDC reference: 1402521
Download citation


Download citation


In the crystal of title molecular salt, the protonated N atom of the 4-methylmorpholin-4-ium cation forms a hydrogen bond with a carbonyl O atom of the barbiturate anion. This N—H⋯O hydrogen bond contributes to the good stability of the reported salt, which exhibits noticeable anticonvulsant and hypnotic activity.
CCDC reference: 1006239
Download citation


Download citation


The crystal structure of seleno-L-cystine, in its hydrochloric acid salt, is isotypic with the common analogue with Se atoms replaced by sulfur, i.e. L-cystine hydrochloride.
CCDC reference: 1403356
Download citation


Download citation


The title compound, an arylsulfonyl glycinyl aryl hydrazone Schiff base, crystallizes with two independent molecules in the asymmetric unit. In the crystal, a series of N—H⋯O and C—H⋯O hydrogen bonds and C—H⋯π and slipped parallel π–π interactions link the molecules, forming a three-dimensional structure.
CCDC reference: 1401257
data reports
inorganic compounds
Download citation


Download citation


metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation




 journal menu
journal menu