issue contents
August 2018 issue
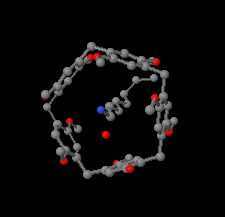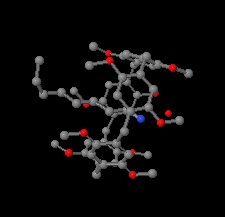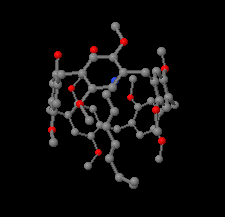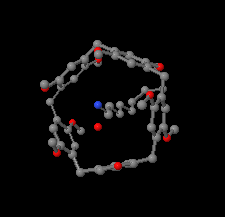
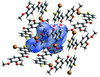
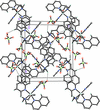
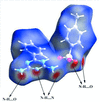
Cover illustration: As a result of their central cavity, monohydroxy pillar[5]arenes can act as hosts for different kinds of guest molecules. This report describes the molecular and crystal structure of the host-guest complex with 1-octanamine. The complex is stabilized by C-H O hydrogen bonding and C-H
O hydrogen bonding and C-H
![[pi]](/logos/entities/pi_rmgif.gif) interactions between the guest and host molecules. A water molecule engages in O-H
interactions between the guest and host molecules. A water molecule engages in O-H N and O-H
N and O-H O hydrogen bonds with the amino and hydroxy groups of the guest and host, respectively, and mediates the formation of dimers. See: Al-Azemi, Vinodh, Mohamod & Alipour [Acta Cryst. (2018). E74, 1117-1120].
O hydrogen bonds with the amino and hydroxy groups of the guest and host, respectively, and mediates the formation of dimers. See: Al-Azemi, Vinodh, Mohamod & Alipour [Acta Cryst. (2018). E74, 1117-1120].
research communications
Download citation


Download citation


In this hydrated 2,6,13,17-tetraazoniatricyclo[16.4.0.07,12]docosane tetrachloride salt, the cation lies about an inversion center. In the crystal, N—H⋯Cl, O–H⋯Cl and N—H⋯O hydrogen bonds connect the anions, cations and water molecules, forming a three-dimensional network.
CCDC reference: 1852146
Download citation


Download citation


A novel mononuclear nickel complex coordinated by 2-(2-hydroxyethyl)pyridine has been synthesized and structurally characterized by X-ray diffraction techniques and photoluminescence spectroscopy. TDDFT calculations have been performed to rationalize the structure explored for this and a related polymeric Cu complex.
CCDC reference: 1481640
Download citation


Download citation


The title compounds both contain a central metal atom in a distorted octahedral geometry coordinated equatorially by four oxygen atoms from water molecules. In the MnII complex, the axial positions are occupied by a nitrogen atom from the ligand and an oxygen atom from the sulfate anion, whereas in the CdII complex they contain two nitrogen atoms from two different ligands and the sulfate anion only serves as the charge-balancing ion. π–π stacking between pyridine rings plays a crucial role in the molecular self-assembly of the two structures.
Download citation


Download citation


The structure of a GaIII complex compound with pentetic acid is reported. The complex molecule is a zwitterion and the GaIII centre is bound in a slightly distorted octahedral coordination sphere by two amine N atoms, three carboxylate O atoms and one water O atom.
CCDC reference: 1852608
Download citation


Download citation


The title compounds, (CH2)nC3H2O(COOH)2 (n = 4, 5), display intermolecular hydrogen bonding, forming a two-dimensional framework.
Download citation


Download citation


The title compound is constructed from two aromatic rings (3-bromo-4-fluorophenyl and 3,4-dimethoxyphenyl), which are linked by a C=C—C(=O)—C enone bridge and form a dihedral angle of 17.91 (17)°. In the crystal, molecules are linked by C—H⋯O hydrogen bonds enclosing rings of 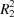 (14) graph-set motif to form layers parallel to (10
(14) graph-set motif to form layers parallel to (10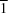 ).
).
CCDC reference: 1852842
Download citation


Download citation


L-proline crystallized, in its zwitterionic form, without the inclusion of any solvent or water molecules through the slow diffusion of diethyl ether into a saturated solution of L-proline in ethanol. In the crystal, the molecules are linked via N—H⋯O hydrogen bonds, resulting in a two-dimensional network.
CCDC reference: 1852963
Download citation


Download citation


The 2-ethylphenyl group in the title compound is disordered over two sets of atomic sites and the molecules are linked into a three-dimensional array by a combination of C—H⋯O hydrogen bonds and nitro⋯π(arene) interactions.
CCDC reference: 1853291
Download citation


Download citation


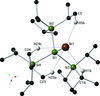
The crystal structure of [Mn(TMPA)(Ac)(CH3OH)]BPh4 (TMPA = tris(pyridin-2-ylmethyl)amine, Ac = acetate, BPh4 = tetraphenylborate) has been determined. The structure reveals a seven-coordinate MnII center with distorted, pentagonal bipyramidal geometry.
CCDC reference: 1853486
Download citation


Download citation


The title Schiff base compound displays a trans configuration with respect to the C=N bond, with the two benzene rings being inclined to each other by 31.90 (12)°.
CCDC reference: 1852926
Download citation


Download citation


The absolute configurations of the 10 asymmetric carbons involved in the structure of the title limonoid, TS3, have been confirmed by resonant scattering. The roles of the water molecules and the C—H⋯π interactions in the crystal packing are highlighted.
CCDC reference: 1854616
Download citation


Download citation


The title compounds were synthesized using the Claisen–Schmidt condensation method and characterized by UV–Vis spectroscopy. The geometrical parameters optimized using density functional theory (DFT) calculations show a good correlation with the experimental results. The small HOMO–LUMO energy gaps of 3.11 and 3.07 eV enhances the non-linear responses of these molecular systems.
Download citation


Download citation


The 5-iodo- and 5-ethynyl-substituted dimethyl isophthalates show molecular frameworks with methyl carboxylate moieties being tilted or perfectly planar with respect to the benzene ring, respectively. Crystal structures feature a three- or two-dimensional supramolecular aggregation in the iodo and ethynyl derivatives, respectively, supported by C—H⋯I and C—H⋯O hydrogen bonding as well as I⋯O and π–π interactions.
Download citation


Download citation


In the complex cation of the title compound, two RuII atoms are bridged via the carbon and oxygen atoms of an anionic CO22– carbonite ligand, resulting in an unsymmetrical dinuclear structure.
CCDC reference: 1855027
Download citation


Download citation


Three 3-arylmethyl-2-[(E)-2-(furan-2-yl)vinyl]-2,3-dihydroquinazolin-4-ones – the products of a three-component reaction between isatoic anhydride, amine and furyl-2-methylacrylaldehyde were studied by X-ray diffraction.
Download citation


Download citation


The two IrIII atoms in the title structure have distorted octahedral coordination spheres, being C,N-chelated by two main 2-phenylpyridine ligands and N,O-chelated by one ancillary 2-[(2,4-dimethoxyphenylimino)methyl]phenolato ligand.
CCDC reference: 1846724
Download citation


Download citation


2-Aminopyridine and citric acid mixed in 1:1 and 3:1 ratios in ethanol yielded crystals of two 2-aminopyridine citric acid salts, viz. C5H7N2+·C6H7O7− (I) and 3C5H7N2+·C6H5O73− (II). Salt I is formed by the protonation of the pyridine N atom and deprotonation of the central carboxylic group of the acid, while in II all three carboxylic groups of the acid are deprotonated and the charges are compensated for by three 2-aminopyridinium cations.
Download citation


Download citation


The crystal structure and supramolecular interactions between a hydroxy-functionalized pillarene and 1-octanamine guest molecule, which forms an interesting host–guest system, are reported.
CCDC reference: 1855261
Download citation


Download citation


The title compound, [Ni(C14H12BrN2O)2][Ni(C13H12BrN2O)2](ClO4)2 2(MeOH) consists of two mononuclear ([Ni(HL)2]2+ and [NiL2]) complexes linked by strong hydrogen bonding [O⋯O separations of only 2.430 (5) Å], which is the shortest reported to date for such species.
CCDC reference: 1856226
Download citation


Download citation


In the crystal structure of the title sulfonamide, intermolecular N—H⋯O hydrogen bonds are present between sulfonamide groups, as well as offset π–π interactions.
CCDC reference: 1856234
Download citation


Download citation


The molecular and crystal structures of 2-iodo benzamide and 2-iodo-N-phenylbenzamide are reported. In both crystals, N—H⋯O hydrogen bonds and C—I⋯π(ring) interactions stabilize the packing with additional C—H⋯π(ring) contacts found in the latter.
Download citation


Download citation


In the crystal, the molecules are linked by weak C–H⋯F hydrogen bonds into the supramolecular inversion dimers.
CCDC reference: 1036795
Download citation


Download citation


The crystal structure of [Cu(DQMEA)(CH3CN)](ClO4)2 (DQMEA = N,N-bis(2-quinolylmethyl)methoxyethylamine) has been determined. The structure reveals a five-coordinate CuII center with a distorted square-pyramidal geometry.
CCDC reference: 1856400
Download citation


Download citation


The title compounds, I (HypSiBr), and II (TipSiBr), crystallized in the cubic space group Pa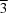 and the triclinic space group P
and the triclinic space group P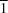 , respectively. In both structures the central silicon atom is sterically hindered with bond angles that deviate from the expected 109.5° of a tetrahedron.
, respectively. In both structures the central silicon atom is sterically hindered with bond angles that deviate from the expected 109.5° of a tetrahedron.
Download citation


Download citation


The title molecule adopts the phenol–imine form. Two intramolecular O—H⋯N hydrogen bonds each generate an S(6) ring motif. In the crystal, weak C—H⋯O hydrogen bonds link the molecules into inversion dimers.
CCDC reference: 1850298
Download citation


Download citation


In the structure of C16H28N2SeHgCl2 the mercury atom is coordinated through two chloride anions, a nitrogen atom and a selenium atom to make up an unusual HgNSeCl2 coordination sphere with an additional long Hg⋯N interaction.
CCDC reference: 1814426
Download citation


Download citation


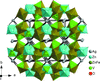
The transition metal orthovanadate, Ag2Zn2Fe(VO4)3, crystallizes in an alluaudite-type structure. ZnII and FeIII atoms are statistically occupied on one general site.
CCDC reference: 1857879
Download citation


Download citation


The asymmetric unit consists of a 2,4-diamino-6-phenyl-1,3,5-triazin-1-ium cation and a 4-methylbenzoate anion. The protonated nitrogen and amino group nitrogen atoms are involved in hydrogen bonding with the sulfonate oxygen atoms through a pair of intermolecular N—H⋯O hydrogen bonds. The inversion-related molecules are further linked by four N—H⋯O intermolecular interations to produce a complementary DDAA hydrogen-bonded array. Hirshfeld surface analysis was employed to further examine the intermolecular interactions.
CCDC reference: 1820866
Download citation


Download citation


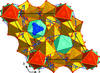
The crystal structures of hydrothermally synthesized TlGa2As(HAsO4)6, CsGa2As(HAsO4)6 and CsAl2As(HAsO4)6 were solved by single-crystal X-ray diffraction. They all crystallize in the RbAl2As(HAsO4)6 structure type (R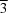 c). The three compounds contain AsO6 octahedra assuming the topological role of M3+O6 octahedra.
c). The three compounds contain AsO6 octahedra assuming the topological role of M3+O6 octahedra.
Download citation


Download citation


In the crystal, the cations and anions stacked along the b-axis direction are linked by C—H⋯Br and N—H⋯Br hydrogen bonds, forming a three-dimensional network. In addition, weak C—H⋯π (ring) interactions, which only involve the minor disorder component. Inversion-related Cl⋯Cl halogen bonds and C—Cl⋯π (ring) contacts also help to stabilize the packing.
CCDC reference: 1857411
Download citation


Download citation


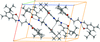
Two deprotonated molecules of Z–Gly–Aib− form a complex with one CaII ion, which assumes a distorted octahedral conformation, whereas the respective metal-free, neutral and symmetry-inverted dipeptides Z–Gly–Aib–OH are mutually hydrogen-bonded.
Download citation


Download citation


The molecular conformation is stabilized via intramolecular C—H⋯O and C—H⋯N contacts. The supramolecular structure is mainly governed by C—H⋯N hydrogen-bonded centrosymmetric dimers, C—H⋯O and C—H⋯S hydrogen bonds and S⋯π stacking interactions, which together lead to the formation of a layered crystal packing.
CCDC reference: 1850683
Download citation


Download citation


The crystal structure of the square-antiprismatic complex tetrakis(1,1,1,5,5,5-hexafluoroacetylacetonato)hafnium(IV) is reported.
CCDC reference: 1857253

 journal menu
journal menu