issue contents
April 2015 issue
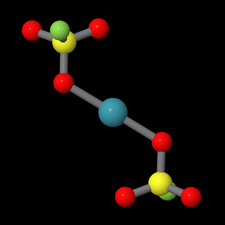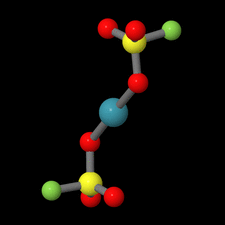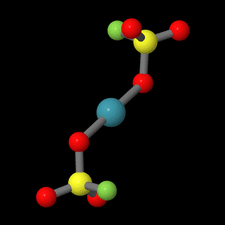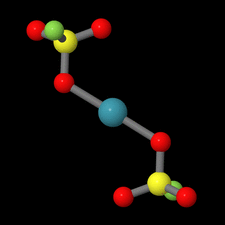
Cover illustration: Xe(SO3F)2 is one of the few examples of a noble gas compound and was synthesized by reacting solid XeF2 with fluorosulfuric acid. Although Xe(SO3F)2 has been known for more than four decades, the molecular and crystal structure remained unclear due to the high thermal instability of this compound. In contrast to earlier assumptions where Cs symmetry was discussed for the molecule in the solid state on the basis of Raman spectroscopic data, the true symmetry is C1, approaching Ci symmetry. This pseudo-inversion symmetry is reflected by the nearly linear O-Xe-O angle of 179.13 (4)° of the two-coordinate xenon atom. See: Malischewski & Seppelt [Acta Cryst. (2015). E71, 363-365].
research communications








































Crystal structure of catena-poly[[aquabis(4-formylbenzoato)-κ2O1,O1′;κO1-zinc]-μ-pyrazine-κ2N:N′]














data reports
metal-organic compounds


























organic compounds


























































 journal menu
journal menu