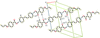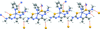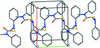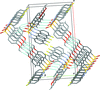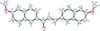issue contents
November 2015 issue
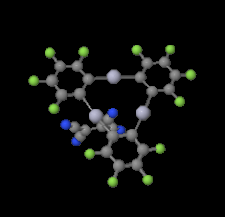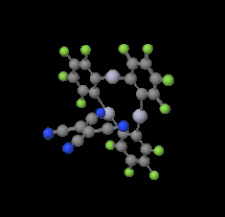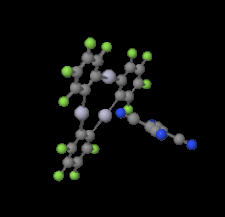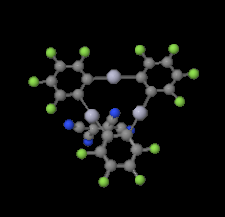
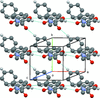
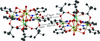
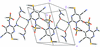
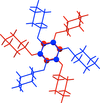
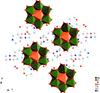
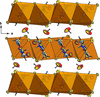
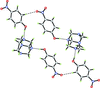
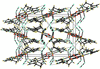
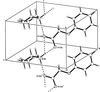
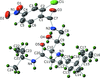
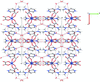
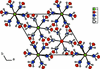
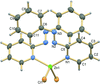
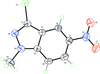
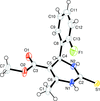
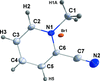
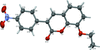
Cover illustration: Trimeric perfluoro-o-phenylene mercury is a macrocyclic Lewis acidthat can be used for the preparation of charge-transfer complexes with unusual optical properties. In combination with the unstable dienophilic compound tetracyanoethylene, this macrocycle reacted to the adduct [Hg3(C6F4)3]·C6N4. In the crystal structure, the macrocycle exhibits point group symmetry 2 and the tetracyanoethylene molecule point group symmetry ![]() . The latter is nearly perpendicular to the mean plane of the macrocycle at a dihedral angle of 88.20 (5)°. Intermolecular interactions between the moieties are weak with Hg
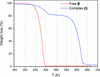
. The latter is nearly perpendicular to the mean plane of the macrocycle at a dihedral angle of 88.20 (5)°. Intermolecular interactions between the moieties are weak with Hg N distances to the nitrile N atoms from the three mercuryatoms of the macrocycle ranging from 2.990 (4) to 3.030 (4) Å Thermogravimetrical analysis revealed that upon complexation tetracyanoethylene is stable to higher temperatures compared to the free state. See: Castañeda, Timofeeva & Khrustalev [Acta Cryst. (2015). E71, 1375-1378].
N distances to the nitrile N atoms from the three mercuryatoms of the macrocycle ranging from 2.990 (4) to 3.030 (4) Å Thermogravimetrical analysis revealed that upon complexation tetracyanoethylene is stable to higher temperatures compared to the free state. See: Castañeda, Timofeeva & Khrustalev [Acta Cryst. (2015). E71, 1375-1378].
research communications






































































data reports
inorganic compounds


metal-organic compounds






















organic compounds











































































































 journal menu
journal menu