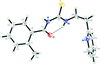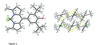issue contents
September 2015 issue
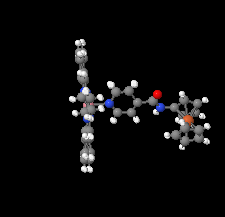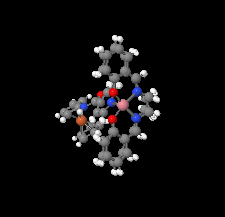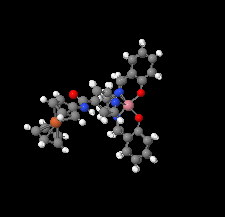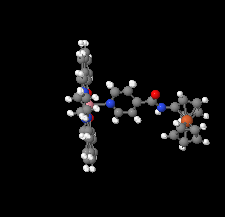
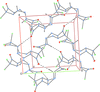
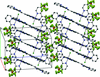
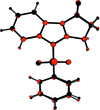
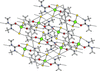
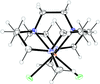
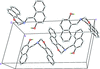
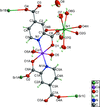
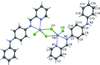
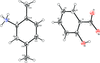
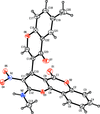
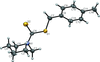
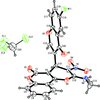
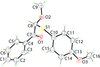
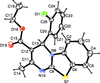
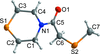
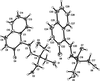
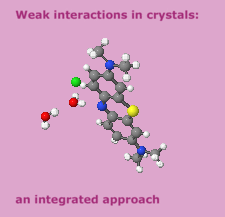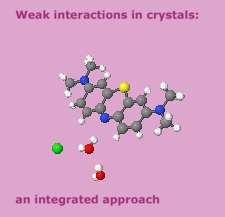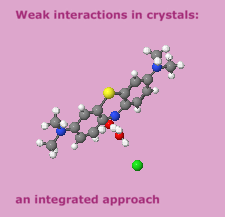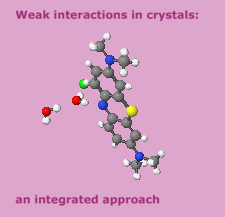
Cover illustration: [CoFe(C5H5)(C16H14N2O2)(C11H9N2O)] combines the functionalities of ferrocene and Co(salen) complexes and may serve as a model compound for oxygen transport. The compound crystallized as disordered ![[lambda]](/logos/entities/lambda_rmgif.gif) and
and ![[delta]](/logos/entities/delta_rmgif.gif) conformers, but a careful analysis of the diffraction data allowed the different salen ring conformations to be established (almost planar in the
conformers, but a careful analysis of the diffraction data allowed the different salen ring conformations to be established (almost planar in the ![[lambda]](/logos/entities/lambda_rmgif.gif) conformer and puckered in the
conformer and puckered in the ![[delta]](/logos/entities/delta_rmgif.gif) conformer). See: Brautigam, Herholdt, Farnsworth, Brudi, McDonald, Wu & Contakes [Acta Cryst. (2015). E71, 1100-1104].
conformer). See: Brautigam, Herholdt, Farnsworth, Brudi, McDonald, Wu & Contakes [Acta Cryst. (2015). E71, 1100-1104].
research communications
Download citation


Download citation


The asymmetric unit contains two independent molecules, each consisting of an eight-membered ring with two four-membered rings fused on either side.
CCDC reference: 1415865
Download citation


Download citation


The non-H atoms in the molecules of three closely-related 4-amino-3-methyl-1H-1,2,4-triazole-5-thiones are either exactly or very nearly co-planar, and the compounds exhibit hydrogen-bonded supramolecular assembly in two, one or zero dimensions.
Download citation


Download citation


The whole molecule of the title Schiff base is generated by twofold rotational symmetry. It crystallizes as a bis-zwitterion, and there are two intramolecular N—H⋯O hydrogen bonds present. In the crystal, molecules are linked by pairs of C—H⋯O hydrogen bonds forming ribbons propagating along [001].
CCDC reference: 1416064
Download citation


Download citation


The crystal structure of enantiopure (3aS,4S,5R,7aR)-2,2,7-trimethyl-3a,4,5,7a- tetrahydro-1,3-benzodioxole-4,5-diol shows that the absolute configuration determined from the synthesis pathway agrees with that determined by X-ray analysis.
CCDC reference: 1416687
Download citation


Download citation


In this first crystal structure of an Ru complex with 6′,6"-bis(pyridin-2-yl)-2,2′:4′,4":2",2"'-quaterpyridine, a `half' of the ligand (one of the two terpyridyl units) is N^N^N mer-coordinated, whereas the other is free and adopts a trans,trans conformation about the interannular C—C bonds. The crystal packing features π–π stacking interactions between tpy–tpy ligands.
CCDC reference: 1416756
Download citation


Download citation


The title compound, [Cd(C5H2N5)2(C3H7NO)2]n, is a two-dimensional coordination polymer extending parallel to (100). Notably, both the primary amino group and the cyano groups are involved in hydrogen-bonding interactions with DMF ligands to direct the assembly and stabilize the crystal packing.
CCDC reference: 1416545
Download citation


Download citation


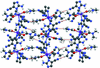
A pentacoordinated Zn2+ ion is found in each independent complex molecule of the title compound; the asymmetric unit is completed by three conformationally flexible non-coordinating benzylamine molecules. Supramolecular layers sustained by N—H⋯N and N—H⋯π interactions are found in the crystal packing; these are connected by π–π contacts.
CCDC reference: 1415614
Download citation


Download citation


A novel hexamolybdate(VI) polyanion protonated by an extra H atom is an unexpected polyanion species among the B-series Anderson-type polyoxometalates (POMs). The extra (seventh) H atom does not lie on a crystallographic centre of symmetry, but is located at the mid-point between two μ2-O atoms of adjacent polyanions, and forms a very short hydrogen bond.
CCDC reference: 1417467
Download citation


Download citation


The title compounds, (I), (II) and (III), are indole derivatives. Compounds (I) and (II) present two independent moieties in the asymmetric unit, and their packing is led by C—H⋯O hydrogen bonds and C—H⋯π interactions. In compound (III), the C—H⋯O hydrogen bonds form 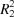 (22) inversion dimers.
(22) inversion dimers.
Download citation


Download citation


In the title compound, the planes of the phenoxy and phthalonitrile rings are oriented at a dihedral angle of 60.39 (5)°. In the crystal, molecules are linked by O—H⋯O, O—H⋯N and N—H⋯O hydrogen bonds, forming slabs parallel to (100). The slabs are linked by a pair of inversion-related C—H⋯N hydrogen bonds, forming a three-dimensional structure.
CCDC reference: 1418026
Download citation


Download citation


In the title compound, there are intermolecular S=O⋯N(nitro) interactions, with an O⋯N distance of 2.9840 (18) Å, between inversion-related molecules. The aromatic rings attached to the SO3 group are oriented in a gauche fashion around the ester S—O bond, with a C—S—O—C torsion angle of 84.68 (11)°.
CCDC reference: 1418463
Download citation


Download citation


Three N-(aryl)-2,2,2-tribromoacetamides show different weak interactions in their crystal structures.
Download citation


Download citation


The CrIII ion in the title cationic complex is coordinated by four N atoms from the macrocyclic ligand, one water molecule and one chloride in a cis geometry, displaying a distorted octahedral environment. The crystal packing is stabilized by N—H⋯Cl, O—H⋯Cl and O—H⋯O hydrogen bonds.
CCDC reference: 1419197
Download citation


Download citation


The geometry around silver(I) metal atom in the title complex is distorted square planar with two normal Ag—N bonds and two long Ag—N bonds.
CCDC reference: 1419095
Download citation


Download citation


The title compound, [Hg(C3H8N2S)2Cl2], is isotypic with its Zn and Cd analogues, having the transition metal in a distorted tetrahedral Cl2S2 coordination environment.
CCDC reference: 1419298
Download citation


Download citation


The tetrahedral tetrachloridocobaltate(II) anion is linked to bis(2-methyl-1H-imidazol-3-ium) cations through N—H⋯Cl hydrogen bonds, resulting in a layered arrangement parallel to (100).
CCDC reference: 1415257
Download citation


Download citation


The RuII atom in the title complex is surrounded by a distorted Cl2N4 coordination set. In the crystal structure, adjacent complex molecules are connected through C—H⋯Cl hydrogen-bonding interactions into a layered arrangement parallel to (100). Additional C—H⋯Br hydrogen-bonding interactions along with π–π stacking interactions complete a three-dimensional supramolecular network.
CCDC reference: 1419653
Download citation


Download citation


Synthesis and structural characterization of a new gold(I) complex with di-2-pyridyl ketone phenylthiosemicarbazone, [AuCl(C18H16N5S)]Cl·1.5H2O
CCDC reference: 1419509
Download citation


Download citation


In contrast to other similar compounds, [FeCl2(C14H30N4)]PF6 is a monomer. Comparison with the mononuclear Fe2+ complex of the same ligand shows that the smaller Fe3+ ion is more fully engulfed by the cavity of the bicyclic ligand. Comparison with the μ-oxo dinuclear complex of an unsubstituted ligand of the same size demonstrates that the methyl groups of 4,11-dimethyl-1,4,8,11-tetraazabicyclo[6.6.2]hexadecane prevent dimerization upon oxidation.
CCDC reference: 1419250
Download citation


Download citation


[Cr(μ-mdea)Cu(μ-Hmdea)(NCS)2H2O], (where mdeaH2 is N-methyldiethanolamine) is formed as a neutral heterometal CuII/CrIII complex whose molecular structure is based on a binuclear {CuCr(μ-O)2} core. In the crystal, the binuclear complexes are linked via two pairs of O—H⋯O hydrogen bonds to form inversion dimers, which are arranged in columns parallel to the a axis.
CCDC reference: 1419706
Download citation


Download citation


In the solvated dinuclear complex [Cu2Br2(ATU)(dppm)2]·2CH3CN, both Cu+ ions adopt distorted tetrahedral geometries, being coordinated by one terminal Br atom, one μ2-S atom of the bridging ATU ligand and two P atoms of the bridging dppm ligands. Within the complex, intramolecular C—H⋯S, C—H⋯π, N—H⋯Br and π–π stacking interactions are observed. In the crystal, the components are linked by N—H⋯Br and C—H⋯N hydrogen bonds and weak π–π stacking interactions, generating chains propagating in the [100] direction.
CCDC reference: 1419827
Download citation


Download citation


The title compound, C13H10N2O7S, was solved in the orthorhombic space group Pna21. The aromatic substituents on the sulfonate group are oriented gauche to one another with a C—O—S—C torsion angle of −62.0 (3)°. The supramolecular features that contribute to the crystal lattice are offset π-π and multiple C—H⋯O interactions.
CCDC reference: 1419864
Download citation


Download citation


A novel naphthoxazine has been synthesized and structurally characterized. In the crystal, pairs of inversion-related molecules are linked into inversion dimers via C—H⋯π interactions.
CCDC reference: 1419687
Download citation


Download citation


Six closely related (2E)-3-aryl-1-(5-halogenothiophen-2-yl)prop-2-en-1-ones all have nearly planar molecular skeletons. C—H⋯O hydrogen bonds are present in only three of the structures but short Br⋯Br, Br⋯O and Cl⋯Cl contacts are also present in some of the structures.
Download citation


Download citation


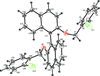
The title cobalt(II) complex was prepared by mixing equimolar amounts of {2,2′-[ethane-1,2-diylbis(nitrilomethanylylidene)]diphenolato}cobalt(II) and N-ferrocenylisonicotinamide in dry dichloromethane under nitrogen and subsequently characterized by ESI–MS, IR, and single-crystal X-ray diffraction. The structure at 100 K has triclinic (P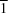 ) symmetry and indicates that the complex crystallizes as a mixture of λ and δ conformers. It exhibits the expected square-pyramidal geometry about the CoII atom, and forms hydrogen-bonded dimers through the amide N—H group and one of the phenolate O atoms on adjacent molecules.
) symmetry and indicates that the complex crystallizes as a mixture of λ and δ conformers. It exhibits the expected square-pyramidal geometry about the CoII atom, and forms hydrogen-bonded dimers through the amide N—H group and one of the phenolate O atoms on adjacent molecules.
CCDC reference: 1417154
Download citation


Download citation


In the packing of the title compound, Ni(H2O)6 is acting as a glue between neighbouring zwitterionic quinoline derivatives which are not directly complexing with NiII.
CCDC reference: 1419884
Download citation


Download citation


Single crystals of CaIrO3 were grown from a CaCl2 flux at atmospheric pressure and crystallized with the post-perovskite type of structure. The crystal structure is reinvestigated on the basis of single-crystal X-ray diffraction data measured using a high-power X-ray source, and the atomic thermal vibration behavior is discussed in terms of the coordination environments.
CCDC reference: 1419830
Download citation


Download citation


The structure of bis(3-bromomesityl)8-quinolyliniumboron(III) tribromide is reported: the refinement indicates that a degree of `over-bromination' of the cation has occurred.
CCDC reference: 1419502
data reports
metal-organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


organic compounds
Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation


Download citation



 journal menu
journal menu